You have shortlisted a Chinese blister packaging machine. The price is compelling — perhaps 40–60% below an equivalent European-branded line. The supplier’s brochure shows CE marks, a glossy ATEX certificate, and the words “EU GMP compliant” printed in bold. Before you countersign that purchase order, ask yourself one critical question: does that machine comply with EU Annex 1 and ATEX Directive 2014/34/EU at the engineering level — or only at the documentation level?
For European pharmaceutical manufacturers operating under the revised EU GMP Annex 1 (2022 edition) and any facility processing solvent-based formulations or handling flammable cleaning agents, these two regulatory frameworks are not optional checkboxes. They define the minimum engineering architecture your packaging line must embody before a single blister can be produced. The consequences of getting this wrong range from failed pre-approval inspections by the EMA or national competent authorities, to full production shutdowns, product recalls, and — in ATEX non-compliance scenarios — catastrophic explosion risk. The stakes could not be higher.
🔥 Forester’s Insight: A 20-Year Engineer’s Perspective
- The Direct Answer: A Chinese blister machine can fully satisfy EU Annex 1 and ATEX requirements — but only if your supplier engineered compliance in from day one, not bolted it on as an afterthought.
- The Field Experience: In my 20 years commissioning lines across Germany, Poland, and the Netherlands, I have watched European buyers sign contracts with Chinese vendors who proudly handed over an ATEX certificate — only to discover during FAT that the solvent-zone enclosures were ATEX-rated but the control panel wiring topology violated Zone 2 segregation rules entirely. The Annex 1 contamination control strategy looked perfect on paper, yet the machine’s HVAC interface had no validated connection point for the cleanroom pressure cascade. No URS clause had captured it. That buyer lost four months and re-engineered at a cost that exceeded the original machine price.
- The Hidden Trap: Compliance documentation and genuine compliance engineering are two completely different products — and cheap suppliers sell you the former. Before issuing a PO, demand a line-by-line URS review against your site’s contamination control strategy and request ATEX zone drawing sign-off from your own SHE team during the FAT stage. At HIJ, we build Annex 1 and ATEX architecture into the mechanical design brief — not the sales brochure.

GMP-compliant structural design: enclosed drive systems, radius-free internal surfaces, and stainless steel frame — minimum requirements for EU Annex 1 blister packaging equipment.
What EU GMP Annex 1 (2022) Actually Demands from Your Blister Machine
The 2022 revision of EU GMP Annex 1, “Manufacture of Sterile Medicinal Products,” introduced a paradigm shift for contamination control: instead of prescribing specific engineering measures, it mandates a holistic Contamination Control Strategy (CCS). For blister packaging lines — which typically operate in Grade C/D environments and interface directly with sterile-filled or moisture-sensitive primary packs — this means your machine vendor must demonstrate how their equipment design actively contributes to your site-level CCS, not merely avoids violating it.
At the mechanical engineering level, Annex 1 compliance for a pharmaceutical blister machine demands the following non-negotiable design elements. Surface finish and material traceability: all product-contact and near-product surfaces must be manufactured from grade 316L stainless steel (or validated equivalent), with an average surface roughness Ra ≤ 0.8 µm on product-contact faces, and Ra ≤ 1.6 µm on non-contact internal surfaces. Welds must be fully penetrating, continuous, and electropolished. Hidden cavities, dead-legs, and bolt-holes inside the cleanroom envelope are non-compliant by design. Every material certificate (EN 10204 3.1 minimum) must be traceable and supplied as part of the qualification documentation package.
HVAC interface and pressure cascade integrity: Annex 1 Section 4 requires that your cleanroom’s unidirectional airflow and differential pressure cascade are not compromised by machine-generated turbulence. This means your blister machine must have defined and documented exhaust connection points, cleanroom-compatible cable penetration seals rated to your process pressure differential (typically +10 to +15 Pa grade differential), and no drive cabinets or heat-generating electronics installed inside the cleanroom envelope unless they are independently ventilated with exhausted airflow directed outside the classified zone. Machines that route servo drive cooling air into the Grade C space are an Annex 1 violation — and a remarkably common one in cheap imports.
Cleaning and sterilisation compatibility: The CCS must demonstrate that the machine can be cleaned in-place (CIP) or at least cleaned-in-situ using validated SOP-driven protocols. This requires drain points, slope-designed frames (minimum 3° drainage angle on horizontal surfaces), and spray-shadow-free internal geometry. WFI (Water for Injection) compatibility of all gaskets and seals — typically EPDM or PTFE rated to 121°C autoclave cycles — must be verified. Finally, electronic batch record integrity under 21 CFR Part 11 / EU Annex 11 is increasingly audited during Annex 1 inspections: your HMI must deliver 21 CFR Part 11-compliant audit trails with electronic signatures, time-stamps referenced to a secure NTP server, and role-based access control. A machine that saves process parameters to a local CSV file with no access controls does not meet this standard regardless of what the spec sheet says.

Precision heat-sealing station on the HIJ pharma blister line: temperature uniformity ±1°C, dwell time ±0.05 s — validated parameters essential for EU Annex 1 process qualification.
ATEX Directive 2014/34/EU: The Engineering Reality Behind the Certificate
ATEX (ATmosphères EXplosibles) compliance for pharmaceutical blister machinery is triggered whenever the operating environment contains — or may transiently contain — flammable solvent vapours or combustible dust in concentrations above the Lower Explosive Limit (LEL). In a typical blister packaging operation, ATEX relevance arises in three scenarios: (1) solvent-based coating or ink systems used in inline printing stations, (2) isopropanol (IPA) or ethanol-based equipment cleaning protocols conducted with the machine partially energised, and (3) PVC/PVDC forming areas where, under abnormal conditions, degradation products can generate flammable off-gases.
The ATEX Directive requires that equipment installed in a classified zone carry an Ex marking indicating the Equipment Group (Group II for surface industries), Equipment Category (Category 2 for Zone 1, Category 3 for Zone 2), and Gas Group/Temperature Class appropriate to the specific solvent or dust present. For example, if your cleaning protocol uses ethanol (Flash point 13°C, Gas Group IIA, T3 ignition temperature class), every electrical component within the classified zone — motors, solenoids, sensors, limit switches, junction boxes, and cable glands — must be individually rated to IIA T3 minimum, and the zone drawing must segregate the ATEX zone boundary with precision.
At the control architecture level, ATEX Zone 2 compliance also mandates that the machine’s main control panel — if located outside the classified zone — must be positively pressurised relative to the process environment (px or pz protection concept per IEC 60079-2), or alternatively that the zone boundary is defined such that the panel is unambiguously outside it. The zone boundary definition must be validated against worst-case vapour dispersion modelling, not simply drawn as a line around the machine footprint. Your SHE (Safety, Health & Environment) manager and a Notified Body-certified ATEX consultant should review and co-sign the zone drawing before FAT acceptance.
The Multi-Supplier Patchwork Nightmare vs. HIJ’s Turnkey Engineering Approach
Many European pharma buyers attempt to build Annex 1 and ATEX compliance by assembling a patchwork: a Chinese blister machine body from Supplier A, an ATEX-rated control panel engineered by a European integrator (Supplier B), HVAC interface engineering by their own facilities team (Supplier C), and IQ/OQ/PQ documentation written by a local CRO (Supplier D). On paper, this appears to divide risk. In practice, it multiplies it — and here is why.
| Risk Area | Multi-Supplier Approach | HIJ Turnkey Approach |
|---|---|---|
| ATEX Zone Drawing Ownership | No single party owns the complete zone drawing; gaps appear at interface boundaries between suppliers | HIJ delivers a unified, Notified Body-reviewed ATEX zone drawing covering the complete machine assembly |
| Annex 1 CCS Integration | Machine vendor has no knowledge of site CCS; HVAC interface undefined until on-site commissioning | HIJ engineering team reviews your site CCS and URS pre-design; HVAC connection points designed into the mechanical brief |
| IQ/OQ/PQ Documentation | Documentation written retrospectively by CRO with limited machine knowledge; deviations discovered late in qualification | FAT/SAT protocols prepared by HIJ engineers concurrent with machine design; IQ package delivered with machine |
| Accountability at Inspection | “It’s the other supplier’s responsibility” — a phrase no inspector accepts | Single point of accountability: HIJ signs the URS compliance matrix |
| Total Project Timeline | Integration delays typically add 4–8 months vs. original plan | Project Certainty: milestones defined, tracked, and guaranteed at contract stage |
Our Turnkey Packaging Solutions are built on one principle: every engineering decision that affects your GMP compliance, ATEX safety, or validation timeline is owned by a single team — ours. From the initial blister packing machine mechanical specification through to on-site SAT sign-off, you deal with one engineer, one project manager, and one accountability chain.

A HIJ complete blister packing line for pharmaceutical production — from thermoforming to blister-cartoning integration — engineered as a single validated system for EU GMP environments.
Technical Specifications: What to Write Into Your URS for EU Annex 1 & ATEX Blister Machines
A well-constructed User Requirements Specification (URS) is your primary weapon against compliance theatre. Below is a non-exhaustive but engineering-grade URS clause framework developed from 20 years of writing and reviewing URS documents for European pharmaceutical clients.
- Material Specification: All product-contact surfaces: AISI 316L, Ra ≤ 0.8 µm (electro-polished), EN 10204 3.1 certificates supplied. Non-contact internal surfaces: AISI 304, Ra ≤ 1.6 µm.
- Forming Station Temperature Control: Heater plate temperature uniformity ±1.5°C across forming tool surface, validated by calibrated thermal mapping during FAT. Thermocouple calibration certificates traceable to national standards.
- Sealing Station Dwell Time: PLC-controlled dwell time accuracy ±0.05 s, seal temperature ±1°C, seal pressure monitored and alarmed. Seal integrity test (vacuum decay or dye ingress per ISO 11607-1) documented in OQ protocol.
- HVAC Interface: Machine to provide DN100 (minimum) HEPA-filtered exhaust connection point with validated air volume flow rate. Cable penetration seals rated to ΔP = 15 Pa. HVAC interface drawing approved by site Facilities Engineering prior to FAT.
- ATEX Classification: Machine to be designed for installation in ATEX Zone 2 (Gas Group IIA, Temperature Class T3 minimum). Full Ex-marking on all electrical components within the classified zone. Zone drawing reviewed and approved by Notified Body. Pressurised control cabinet (px/pz concept) if panel located within 3 m of the zone boundary.
- Electronic Batch Records: HMI to provide 21 CFR Part 11 / EU Annex 11-compliant audit trail. All parameter changes logged with: operator ID (role-based access), timestamp (NTP-synchronised, UTC), previous value, new value, reason for change (mandatory field). Audit trail data stored in validated, non-editable format. Export function to PDF/A or CSV with digital signature.
- Reject System: 100% blister vision inspection system with automatic rejection. Rejected blisters directed to locked reject container with count reconciliation in electronic batch record. No manual override of rejection without level 3 supervisor electronic signature.
- Cleaning Validation Support: Machine to provide equipment-specific Cleaning Validation Support Package: surface area calculations, material compatibility data for validated cleaning agents (IPA, WFI, dilute caustic), worst-case sampling point locations, recovery study protocol template.
- FAT/SAT Protocol: Supplier to provide draft IQ/OQ protocols minimum 4 weeks prior to FAT. FAT to include: dimensional verification, material certificate review, electrical safety test (EN 60204-1), ATEX component verification against zone drawing, HMI audit trail demonstration, sealing integrity test, OEE measurement at rated speed for minimum 4-hour continuous run.
Cold-Form Alu-Alu vs. Thermoforming: ATEX & Annex 1 Implications Differ Significantly
European buyers sourcing equipment for moisture-sensitive molecules — particularly biologics, peptides, and hygroscopic small molecules — frequently specify Alu-Alu cold-form blister packaging machines, which offer near-zero moisture vapour transmission rate (MVTR). From an Annex 1 and ATEX perspective, cold-form machines present a distinct engineering profile versus thermoforming lines.
Cold-form Alu-Alu machines operate at significantly lower forming temperatures (forming achieved through mechanical pressure rather than heat, typically ambient to 40°C), which reduces the risk of solvent vapour generation from the film itself. However, the cold-form process uses a high-tonnage forming press — typically 80–150 kN — which generates mechanical vibration and requires robust frame rigidity to maintain cleanroom floor load specifications (typically 5 kN/m² for Grade C/D cleanrooms). The forming tool change process must be validated as a cleanroom-compatible procedure: tool changes generate particulate, and without a validated change protocol and post-change surface clean verification, each format change is a potential Annex 1 contamination event. For thermoforming lines, the additional Annex 1 engineering consideration is the forming heater: the heating plate operates at 120–180°C for PVC/PVDC forming, creating a localised heat source that must be modelled in your cleanroom thermal validation to ensure it does not disrupt unidirectional airflow patterns above the product exposure zone.
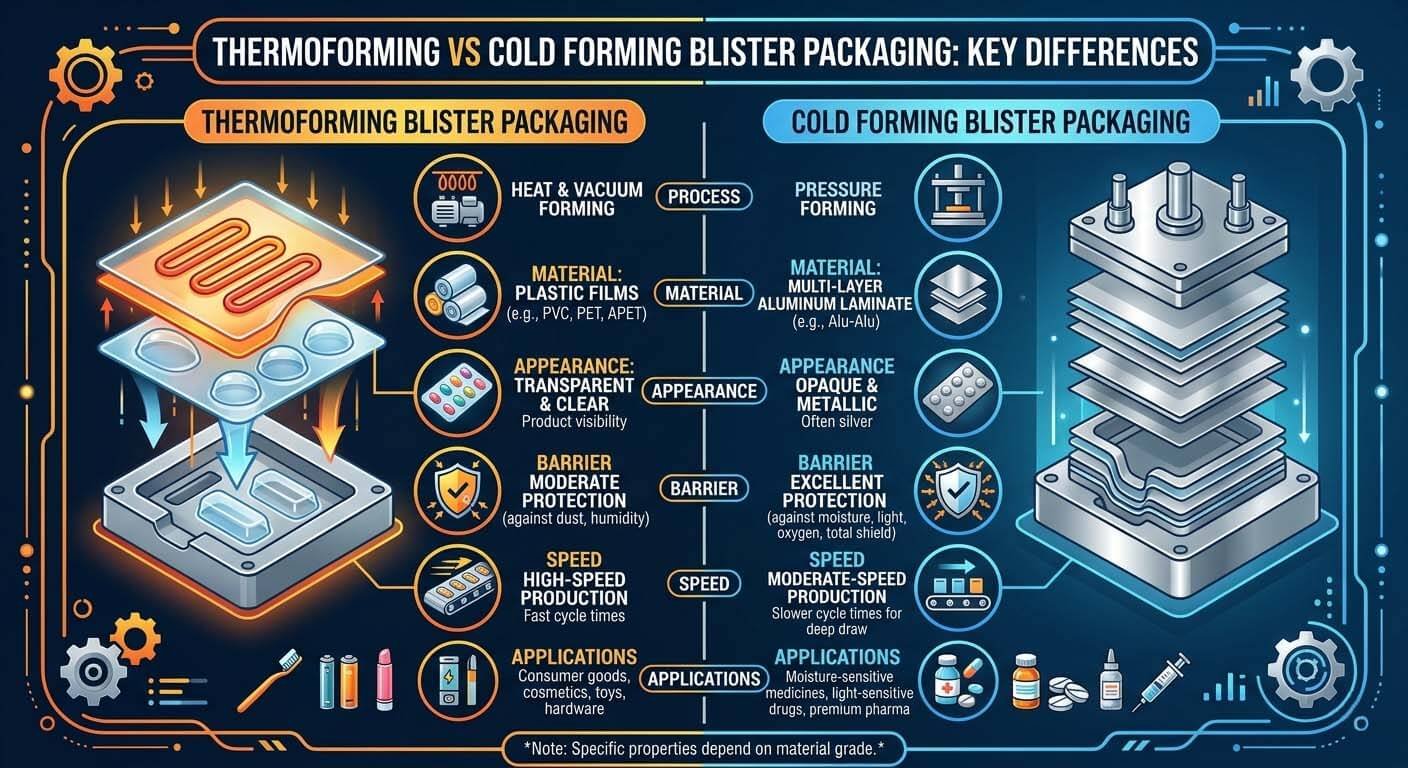
Thermoforming (PVC/PVDC) vs. cold-form (Alu-Alu) blister packaging: the engineering architecture — and Annex 1/ATEX compliance implications — differ significantly between both technologies.
The HIJ DPP-250 Pharma Blister Packaging Machine and the DPP-260 Automatic Blister Packing Machine are both available in EU Annex 1-engineered configurations, with ATEX Zone 2 options. Our engineering team will review your specific ATEX zone classification, solvent identity, Gas Group, and Temperature Class requirements as part of the URS review process — before we issue a commercial proposal. This is not a premium service add-on. It is our standard engineering practice, because we have seen what happens when it is skipped.
HIJ machines integrate globally trusted components for assured compliance and maintainability in European facilities:





Frequently Asked Questions
Q1: Does a CE mark on a Chinese blister machine automatically mean it is EU Annex 1 and ATEX compliant?
No — and this is one of the most dangerous misconceptions in European pharma procurement. The CE mark certifies compliance with the EU Machinery Directive (2006/42/EC) and, where applicable, the Low Voltage Directive (2014/35/EU). It confirms that the machine meets basic safety, EMC, and mechanical design requirements. EU GMP Annex 1 is not a product directive — it is a manufacturing practice standard enforced by your national competent authority during GMP inspections. ATEX compliance (Directive 2014/34/EU) is a separate CE marking pathway requiring involvement of a Notified Body for Category 1 and 2 equipment. A blister machine can carry a legitimate CE mark and still fail Annex 1 qualification or ATEX installation inspection. Always request separate documentation: the ATEX EC-Type Examination Certificate (for Category 2) from a recognised Notified Body (e.g., PTB, DEKRA, SGS), the ATEX as-built zone drawing, and the GMP compliance matrix mapped against your URS.
Q2: What qualifications documentation should a Chinese blister machine supplier provide for European pharma buyers?
At minimum, a supplier targeting European regulated markets should provide: (1) Design Qualification (DQ) package including URS compliance matrix, mechanical drawings, P&ID if applicable, and material certificates; (2) Factory Acceptance Test (FAT) protocol — drafted by the supplier, reviewed by the buyer, and executed at the supplier’s facility with buyer witness; (3) IQ (Installation Qualification) template pre-populated with machine-specific data; (4) OQ (Operational Qualification) protocol including critical process parameter ranges, alarm and interlock verification, and electronic batch record functionality test; (5) Risk Assessment per ICH Q9 / ISO 14971 for critical quality attributes (CQAs) linked to machine design parameters; (6) ATEX documentation pack if applicable: Ex-marking declaration, zone drawing, component certifications; (7) Software validation summary per GAMP 5 category classification. At HIJ, all of the above are included in our standard service and support package — not sold as extras.
Q3: What is the correct ATEX zone classification for a pharmaceutical blister packaging line that uses IPA for routine cleaning?
This depends on IPA usage frequency, volume, and facility ventilation, but the standard industry approach is as follows. During normal blister packaging operation with no IPA present, the immediate machine environment is typically unclassified (Zone NE). During IPA cleaning procedures — which are intermittent and controlled — a Zone 2 classification applies within the immediate machine envelope and a radius defined by your site ATEX explosion protection document (EPD). IPA has a Flash Point of 13°C, belongs to Gas Group IIA, and has an autoignition temperature of 425°C (Temperature Class T3). Therefore, any electrical equipment that remains energised during IPA cleaning must be rated to IIA T3 minimum. The most practical and compliant engineering solution is to de-energise all non-ATEX electrical components before commencing IPA cleaning — a procedure that must be captured in your SOPs and verified during OQ. HIJ can supply machines with an ATEX-compliant “cleaning mode” interlock that enforces this de-energisation sequence automatically and logs the event in the audit trail.
Q4: How does HIJ Machinery ensure its blister machines integrate with a European client’s existing cleanroom pressure cascade?
This is addressed at the URS stage, not at installation. Our engineering process requires the client to provide their site HVAC validation report — specifically the differential pressure cascade map (Grade C/D boundary, typically ΔP 10–15 Pa minimum) and the room air change rate (ACR) specification. Our mechanical designers then model the machine’s thermal and airflow contribution using CFD (Computational Fluid Dynamics) screening, identify any potential disruption to unidirectional airflow or pressure differential maintenance, and specify exhaust connection points and cable penetration seal ratings accordingly. This is not standard practice in the Chinese equipment industry — most suppliers define the machine envelope and leave HVAC integration entirely to the buyer. At HIJ, this integration engineering is part of our scope. Explore our engineering philosophy and compliance commitment to understand why this approach consistently delivers shorter qualification timelines for our European clients.
Conclusion: Delivering Project Certainty for European Pharma
The revised EU GMP Annex 1 and the ATEX Directive 2014/34/EU represent the two most technically demanding compliance frameworks a blister packaging machine must navigate in the European pharmaceutical market. Neither framework is satisfied by a certificate alone. Both require that compliance be engineered into the machine’s design brief — in the mechanical architecture, the electrical topology, the HVAC interface specification, and the software validation structure — before a single component is fabricated.
A Chinese blister machine can absolutely meet these standards. I have designed, built, and commissioned lines that have passed EMA pre-approval inspections and ATEX installation verification at European pharmaceutical sites. But the critical differentiator is this: did the supplier’s engineering team build Annex 1 and ATEX requirements into the design from Day 1, or did the compliance team paste a certificate onto a standard machine at the end of the sales process? After 20 years in this industry, I can tell the difference in the first 10 minutes of a technical review. Your inspector can too.
At HIJ Machinery, I don’t just sell you a machine; I deliver project certainty. Every blister packing machine we supply to a European regulated market comes with a URS compliance matrix signed by our engineering lead, an ATEX zone drawing reviewed before contract, and a FAT protocol agreed before fabrication begins. That is not a promise — it is our standard contract deliverable.
Ready to Validate Your Blister Line for EU Annex 1 & ATEX?
Send us your URS or your site’s contamination control strategy summary. Our engineering team will review your Annex 1 and ATEX requirements, specify the correct machine architecture, and provide a documented Turnkey Quote within 48 hours — with FAT/SAT protocols included.








