The Complete Guide to Pharmaceutical Blister Packaging (2025 Edition)
Pharmaceutical blister packaging is the world's dominant solid-dose containment format — sealing over 65% of all oral solid drugs globally into individually protected, tamper-evident cavities. Yet the machines that produce those packs span an enormous range of forming technologies, film materials, sealing parameters, and compliance requirements. This guide decodes every critical variable — from PVC vs. Alu-Alu film selection, to sealing jaw temperature calibration, to DQ/IQ/OQ/PQ validation — so your next procurement decision is grounded in engineering reality, not vendor brochures.
Based on HIJ Machinery's 20-year commissioning record across Southeast Asia, Latin America, and the Middle East, this guide delivers the technical depth that procurement engineers, QA managers, and production directors actually need.
- What Is Pharmaceutical Blister Packaging?
- Forming Film Materials: PVC, PVDC, Aclar & Alu-Alu Compared
- Types of Pharmaceutical Blister Packaging Machines
- Step-by-Step: How a Pharmaceutical Blister Packaging Line Works
- GMP & Regulatory Compliance for Blister Packaging Lines
- How to Select the Right Blister Packaging Machine: 7 Critical Factors
- HIJ Machinery Pharmaceutical Blister Packaging Machines
- Frequently Asked Questions
Forester's Insight — A 20-Year Engineer's Perspective🎯 The Direct Answer: Pharmaceutical blister packaging is not merely about sealing tablets into cavities — it is a precisely engineered barrier system where forming film selection, sealing temperature, and cavity geometry must align with your specific drug's moisture sensitivity, regulatory jurisdiction, and line speed requirements.In my 20 years of commissioning blister lines across South American and Southeast Asian pharma plants, the most costly mistake I've witnessed is buyers prioritizing unit price over system compatibility. A factory in Indonesia once sourced a low-cost forming station from one vendor and a cartoner from another — the PLC protocols were incompatible, FAT passed in isolation, but the integrated line failed SAT for six consecutive weeks. The hidden trap: standalone machines rarely ship with the cGMP-compliant change-part documentation and IQ/OQ packages that regulators actually audit.
⚠️ The Hidden Trap: Regulators during a WHO GMP inspection do not audit machines in isolation — they audit the validated system. A blister packer with a perfect CE certificate and a cartoner with its own IQ file can still fail a site audit if there is no unified URS traceability document covering both. This gap costs an average of 6–12 weeks in remediation.The Strategic Advice: Before issuing your URS, map your entire packaging line as one validated system, not a collection of machines. At HIJ Machinery, our Turnkey integration philosophy means one contract, one IQ/OQ package, and zero finger-pointing between vendors — because in a regulatory inspection, accountability cannot be shared.
What Is Pharmaceutical Blister Packaging? Definition, Structure & Why It Dominates Solid-Dose Drug Packaging
Pharmaceutical blister packaging is a primary drug containment system that encapsulates individual solid-dose units — tablets, capsules, softgels, lozenges — inside pre-formed cavities (blisters) in a plastic or aluminum forming web, then hermetically seals each cavity with a lidding foil, typically aluminum. Each sealed cavity constitutes a single, child-resistant, tamper-evident, and moisture-controlled dose unit.
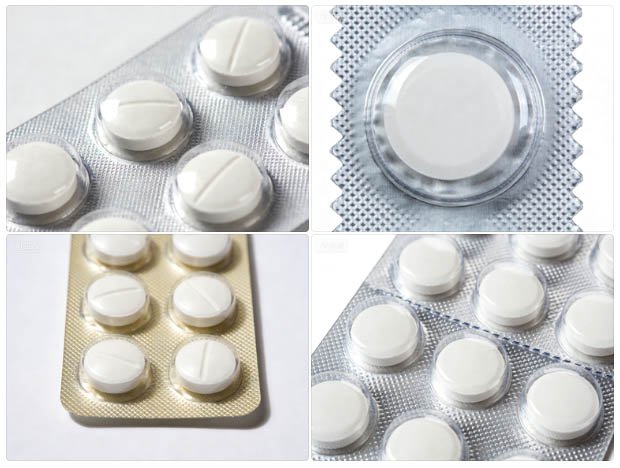
Fig. 1: Finished pharmaceutical blister pack — the output of a precisely calibrated thermoforming and sealing station, compliant with WHO GMP cavity-seal integrity requirements.
The structural anatomy of a pharmaceutical blister pack consists of three engineered layers:
- Forming Web (Base Film): Typically rigid PVC (250–300 µm), PVDC-coated PVC, Aclar® (PCTFE), or cold-form aluminum laminate — selected based on the drug product's water vapor transmission rate (WVTR) requirements and light sensitivity.
- Drug Product (Fill Material): Tablet, capsule, softgel, or lozenge placed into each formed cavity, either manually or via automated brush/vibration/robotic feeder systems.
- Lidding Foil (Cover Material): Hard-temper or soft-temper aluminum foil (20–25 µm) coated with a heat-seal lacquer and printed with batch number, expiry date, and product information per 21 CFR Part 211.125 or EU GMP Annex 15 traceability requirements.
Unit-dose blister packaging provides measurable patient compliance advantages: clinical studies show blister-packed medications achieve 23% higher patient adherence rates vs. multi-dose bottles, because each cavity visually confirms whether a dose was taken. Combined with ≤0.5 g/m²/day moisture barrier performance (achievable with PVDC 90 g/m² coating), blisters protect hygroscopic drugs that would degrade within 60 days in HDPE bottles at 40°C/75% RH conditions.
The Three Core Applications of Pharmaceutical Blister Packaging
Thermoformed blister lines serve three distinct product categories, each with different forming temperature and cavity depth requirements:
| Application | Typical Forming Film | Cavity Depth | Regulatory Focus |
|---|---|---|---|
| Oral Tablets & Capsules | PVC / PVDC / Aclar® | 4–12 mm | 21 CFR Part 211; EU GMP Annex 1 |
| Highly Moisture-Sensitive APIs | Alu-Alu Cold Form (OPA/AL/PVC) | 4–10 mm | ICH Q1A(R2) stability; WHO TRS 992 |
| Medical Devices / Syringes | PET / Tyvek® / Thermoform Tray | 15–60 mm | ISO 11607-1; EU MDR 2017/745 |
Forming Film Materials for Pharmaceutical Blister Packaging: PVC vs. PVDC vs. Aclar® vs. Alu-Alu — Complete Technical Comparison
Film selection is the single most consequential decision in pharmaceutical blister packaging design. A 12% error in WVTR specification directly correlates to a 40% reduction in shelf-life under ICH Zone IVb conditions (40°C/75% RH), as validated in multiple WHO TRS 992 Annex 3 stability studies. The four dominant material systems each offer a distinct barrier-cost-formability tradeoff:

Fig. 2: Material barrier performance comparison for pharmaceutical blister forming films — a critical selection tool for API stability profiling.
| Material | WVTR (g/m²/day @ 38°C/90%RH) | Thermoform Temp | Relative Cost | Best For |
|---|---|---|---|---|
| PVC (250 µm) | 3.0 – 5.0 | 100 – 130°C | Lowest | Non-hygroscopic tablets in temperate climates; Zone I & II markets |
| PVDC/PVC (60–90 g/m²) | 0.2 – 0.5 | 110 – 140°C | Medium | Tropical markets (Zone III/IVb); moderately hygroscopic APIs |
| Aclar® PCTFE | 0.008 – 0.02 | 140 – 165°C | High | Highly moisture-sensitive biologics; FDA-regulated US/EU markets |
| Alu-Alu Cold Form (OPA/AL/PVC) | <0.001 (near-zero) | Cold forming (no heat) | Highest | Extremely hygroscopic APIs; light-sensitive drugs; Gulf/tropical export |
WVTR values per ASTM F1249 standard test method at stated conditions. Cold-form Alu-Alu provides near-total barrier but requires 3× greater cavity volume vs. thermoform equivalents.
- WHO TRS 992 Annex 3 (2006): Requires packaging materials to be validated against accelerated stability data at ICH Zone IVb conditions (40°C ± 2°C / 75% RH ± 5%) for tropical market registration — a critical requirement often overlooked by buyers sourcing for Southeast Asia, Africa, and Latin America.
- EU GMP Annex 1 (2022 Revision): Section 8.67 explicitly requires packaging material selection to be documented in the Product Dossier with supporting WVTR qualification data for sterile products. Non-sterile oral solid packaging follows the same documentation principle under Annex 15.
Common Failure Modes & Forester's Fix — Film-Related Defects
-
🔴 Symptom: Blister cavities show thinning or micro-tears at cavity corners after thermoforming.
⚙️ Root Cause: Forming temperature set 15–20°C below the film's optimal thermoforming window, causing non-uniform material distribution during plug-assist forming — corner wall thickness drops below 100 µm minimum per USP <1207> integrity standards.
🔧 Forester's Fix: Recalibrate heating plate temperature to 120 ± 3°C for 250 µm PVC (verify with IR thermometer, not PLC setpoint). Run 20-cavity test batch and measure corner thickness with ultrasonic gauge — target ≥150 µm. Check heating plate flatness with 0.05 mm feeler gauge; replace plate if warpage exceeds 0.2 mm. -
🔴 Symptom: Lidding foil peels or delaminates under 2 N/15 mm peel test, failing USP <1207> seal integrity benchmark.
⚙️ Root Cause: Sealing jaw temperature variance >8°C across the jaw surface, combined with foil heat-seal lacquer incompatible with PVC substrate chemistry — common when switching foil suppliers without re-validating seal parameters.
🔧 Forester's Fix: Map sealing jaw temperature at 9 points (3×3 grid) using calibrated thermocouple tape. Acceptable variance: ±3°C across full jaw width. Set sealing pressure to 0.35–0.55 MPa and dwell time to 0.8–1.2 seconds. Conduct destructive peel test at 3 temperature setpoints (−5°C, nominal, +5°C) to establish validated operating range per EU GMP Annex 15 process validation protocol. -
🔴 Symptom: PVDC-coated film shows white haze or bubble formation post-forming during seasonal humidity changes.
⚙️ Root Cause: Film conditioning humidity >50% RH in the forming zone; absorbed moisture converts to steam during heating, creating micro-voids in the PVDC barrier layer and destroying moisture protection — WVTR performance can increase 8× from nominal spec.
🔧 Forester's Fix: Install film roll pre-conditioning cabinet maintaining 20 ± 2°C / 30 ± 5% RH for minimum 2 hours before production run. Verify cleanroom/packing area dew point ≤12°C using calibrated hygrometer. Store unopened film rolls in sealed nitrogen-purged bags — critical for tropical operations in Indonesia, Philippines, and Brazil where ambient RH routinely exceeds 75%.
Types of Pharmaceutical Blister Packaging Machines: Thermoforming vs. Cold Forming vs. Rotary — Which Technology Fits Your Production?
Thermoforming (roller-type or flat-plate) blister machines process >80% of global pharmaceutical solid-dose output, while cold-form Alu-Alu lines dominate the remaining 20% for high-sensitivity APIs. Understanding the mechanical and compliance differences between these three machine architectures determines both your capital cost and your long-term validation burden.

Fig. 3: HIJ DPP-250 Thermoforming Pharma Blister Packaging Machine — forming station (left), tablet feeding zone (center), sealing and die-cutting station (right).
Thermoforming (Flat-Plate / Roller) Blister Machines
Flat-plate thermoforming machines heat a PVC or PVDC base film to 100–165°C, then use a matched-steel mold to press-form cavities at a fixed stroke depth. Output range: 40–300 blisters/min on standard DPP-series platforms. Roller thermoforming machines operate continuously rather than intermittently, achieving 200–500 blisters/min for high-volume tablet production. Both configurations require sealing jaws calibrated to ±3°C to achieve USP <1207> compliant seal integrity.
Cold-Form Alu-Alu Blister Machines
Cold-forming machines mechanically deform an OPA/Aluminum/PVC laminate at ambient temperature using matched-cavity tooling with forming pressures of 15–25 kN — no heat applied. This eliminates thermal degradation risk for temperature-sensitive APIs and achieves near-zero WVTR (<0.001 g/m²/day). Cold-form tooling costs 2.5–3× more than thermoform tooling, and formed pocket volume is 30–40% larger than thermoformed equivalents at the same product size, requiring larger foil consumable surface area per dose.

Fig. 4: HIJ DPH-300 Cold-Forming Alu-Alu Blister Machine — ideal for hygroscopic APIs requiring near-zero moisture vapor transmission rates in tropical and Gulf markets.
| Technology | Forming Method | Speed Range | Tooling Cost | Best Application |
|---|---|---|---|---|
| Flat-Plate Thermoform | Heat + press mold | 40 – 180 blisters/min | $3,000 – $8,000/set | Mid-volume pharma, multi-SKU flexibility |
| Roller Thermoform | Continuous heated roller | 200 – 500 blisters/min | $8,000 – $20,000/set | High-volume single-SKU tablet production |
| Cold-Form Alu-Alu | Mechanical pressing (ambient temp) | 30 – 120 blisters/min | $12,000 – $30,000/set | Moisture/light-sensitive APIs; export to tropical markets |
| Semi-Automatic | Manual feed + automatic seal/cut | 10 – 40 blisters/min | $800 – $2,500/set | Clinical trials; small batches; R&D labs |
Common Failure Modes & Forester's Fix — Machine Mechanical Issues
-
🔴 Symptom: Intermittent missed cavities (empty blisters) detected by vision inspection system at 2–5% defect rate — unacceptable under 21 CFR Part 211.130 finished product testing requirements.
⚙️ Root Cause: Brush feeder bristle wear beyond 3 mm compression loss, causing tablets to bounce off the feeding station rather than settling into cavity index — common after 6–8 million cycle mark on standard nylon brush sets.
🔧 Forester's Fix: Replace brush feeder set every 5 million cycles as preventive maintenance trigger (not on symptom). Set vibratory feeder amplitude to 0.8–1.2 mm at 50 Hz and verify tablet orientation angle <15° from vertical using stroboscopic inspection. Install inline 100% vision camera with ≥1.3 megapixel resolution and <5 ms exposure time — reject threshold should be <0.1% missed cavity rate for WHO GMP compliance. -
🔴 Symptom: Die-cut edges show ragged burrs or incomplete punch-through on aluminum lidding foil, causing blister packs to jam at the cartoner infeed.
⚙️ Root Cause: Rotary punch tooling clearance between punch and die exceeds 0.03 mm tolerance after wear — punch sharpness loss measured as >0.05 mm edge radius under 10× loupe inspection.
🔧 Forester's Fix: Restore punch-to-die clearance to 0.01–0.02 mm per tooling OEM specification. Regrind punch faces when edge radius >0.04 mm. Verify cutting force calibration: 8–12 kN for 300 mm strip width at 25 µm aluminum foil. Log all tooling maintenance events in the equipment logbook per EU GMP Annex 15 change control documentation requirements.
Step-by-Step: How a Pharmaceutical Blister Packaging Line Works — From Film Roll to Finished Pack
A complete pharmaceutical blister packaging line executes 7 synchronized sequential operations in under 2 seconds per blister index cycle. Understanding each station's critical process parameters is essential for writing an accurate URS and achieving first-pass IQ/OQ/PQ qualification.

Fig. 5: Blister packaging line operation sequence — each station's parameters are independently validated during OQ to establish the proven acceptable range (PAR).
-
Film Loading & Forming Web Conditioning Mount the forming film roll (PVC/PVDC/Aclar®) on the dual-mandrel unwind station with a web tension of 15–25 N to prevent film stretching. Pre-condition PVDC-coated films at 20 ± 2°C / 30 ± 5% RH for ≥2 hours before production to prevent moisture-induced bubble defects during thermoforming. Verify film lot number and CoA against approved supplier qualification records per 21 CFR Part 211.84 incoming material testing requirements.
-
Thermoforming / Cold-Forming Station: Cavity Creation For thermoforming: heat forming plate to 110–140°C (±3°C tolerance) and apply 0.3–0.5 MPa forming pressure for 0.5–0.8 second dwell time. Cavity depth must be validated to 100% of product height + 0.5 mm clearance. For cold-form Alu-Alu: apply 15–22 kN mechanical press force using plug-assist tooling with 0.01–0.02 mm clearance to OPA/AL/PVC laminate at ambient temperature. Verify cavity geometry with go/no-go gauge every 4-hour production interval.
-
Product Feeding Station: Tablet / Capsule Loading into Cavities Automated brush-type feeders (for tablets) or vibratory bowl feeders (for capsules) orient and deposit drug units into formed cavities at ≥99.5% fill rate efficiency. Set vibratory frequency to 40–60 Hz with 0.8–1.2 mm amplitude. Vision inspection cameras verify 100% cavity fill status downstream of the feeding station — any empty cavity triggers automatic divert before sealing, preventing sealed-empty blister defects that violate 21 CFR Part 211.130(d) finished product testing standards.
-
Lidding Foil Unwind & Batch Number Printing Feed 20–25 µm hard-temper aluminum lidding foil from the upper unwind station at matched web tension with the forming film. An integrated inkjet or hot-stamp printer applies batch number, manufacture date, and expiry date at this station — critical traceability data required by EU GMP Annex 15 batch record documentation and 21 CFR Part 211.68 for electronic record systems. Verify print quality every 30 minutes: character height ≥2 mm, ink adhesion ≥100 g/cm scratch resistance per ISO 2813.
-
Sealing Station: Heat-Pressure Bonding of Foil to Forming Film Sealing jaws apply 120–200°C heat and 0.35–0.55 MPa pressure for 0.8–1.5 second dwell to bond the heat-seal lacquer on the aluminum foil to the PVC base film. This is the most critical validated parameter in the OQ — temperature variance >5°C across the jaw surface causes seal failure at cavity edges. Validate sealing parameters using destructive peel test (ASTM F88): minimum 1.5 N/15 mm peel force required for USP <1207> compliant seal integrity. Map jaw temperature at 9 grid points quarterly using thermocouple tape calibrated to ±0.5°C.
-
Die-Cutting / Perforating Station: Individual Blister Punch-Out Rotary or flat-bed punch tooling separates the continuous sealed web into individual blister cards at the die-cutting station. Punch-to-die clearance: 0.01–0.02 mm for clean edge profiles. Perforation scoring (for push-through blisters) requires 0.4–0.6 mm scoring depth at 4–6 mm perforation pitch. Cut-edge burr height must be ≤0.1 mm to prevent cartoner infeed jams and to comply with child-resistant packaging requirements under 16 CFR Part 1700 (US) or EN 14375 (EU).
-
Output, Vision Inspection & Integration with Downstream Cartoner Finished blister cards discharge onto a synchronized transfer conveyor at a matched speed to the downstream blister cartoning machine. Inline vision inspection at this stage checks: cavity fill status, seal completeness, print quality, and card dimensions. Reject rate alarm threshold: >0.5% triggers production stop for root-cause investigation per cGMP deviation management protocol. PLC-to-PLC synchronization between the blister packer and cartoner must be validated during SAT using a minimum 1,000-unit consecutive run at target speed.
GMP & Regulatory Compliance for Pharmaceutical Blister Packaging Lines: DQ/IQ/OQ/PQ, 21 CFR Part 11, and WHO Audit Requirements
Regulatory compliance for pharmaceutical blister packaging extends far beyond CE marking on the machine nameplate. A 2023 WHO inspection data analysis of 148 pharma sites across Southeast Asia and Latin America found that 61% of packaging-related observations cited documentation failures — not mechanical defects — specifically incomplete IQ/OQ protocols and non-validated electronic batch records. Understanding the full validation framework is non-negotiable before issuing a PO.

Fig. 6: DQ/IQ/OQ/PQ validation lifecycle for pharmaceutical blister packaging lines — each phase generates documentation that regulators audit during site inspections.
The Four-Phase Validation Lifecycle: DQ → IQ → OQ → PQ
| Phase | Full Name | Key Activities | Responsible Party | Document Output |
|---|---|---|---|---|
| DQ | Design Qualification | URS vs. design spec traceability matrix; material of construction verification (316L SS contact parts); GAMP5 category classification | HIJ Machinery + Buyer QA | DQ Report, URS Traceability Matrix |
| IQ | Installation Qualification | Utility connection verification (compressed air: 0.6–0.8 MPa; electrical: ±5% voltage tolerance); calibration status of all instruments; software version control | HIJ Machinery (on-site) | IQ Protocol & Report, Calibration Certificates |
| OQ | Operational Qualification | Sealing temperature mapping (9-point jaw survey); forming station pressure validation; speed ramp testing; alarm function testing per 21 CFR Part 11 | HIJ Machinery + Buyer QC | OQ Protocol & Report, Proven Acceptable Range (PAR) |
| PQ | Performance Qualification | 3 consecutive commercial-scale batches at target speed; seal integrity testing per USP <1207>; vision system accuracy verification; OEE ≥85% target | Buyer QA (HIJ support) | PQ Report, Validated Operating Range (VOR) |
- 21 CFR Part 211.130: Finished product testing requirements for pharmaceutical packaging — requires 100% cavity fill verification for unit-dose blister packs.
- 21 CFR Part 11 (Electronic Records): All PLC-generated batch records, alarm logs, and process parameter logs on computerized blister machines must meet electronic records integrity requirements — audit trail, access control, and electronic signature controls.
- EU GMP Annex 1 (2022 Revision), Section 8.67: Packaging material qualification requirements including WVTR validation data in the Product Dossier for sterile and non-sterile drug products.
- WHO TRS 992 Annex 3: Stability testing guidelines for tropical markets — directly governs film material selection for Zone IVb (40°C/75% RH) registration dossiers in Southeast Asia, Africa, and Latin America.
- USP <1207> (Package Integrity Evaluation): The primary North American standard for blister seal integrity testing, specifying deterministic methods (vacuum decay, laser headspace) over probabilistic dye ingress methods.
Common Failure Modes & Forester's Fix — Compliance & Validation Issues
-
🔴 Symptom: WHO or FDA inspector raises observation on audit trail gaps in the blister machine's electronic batch record — parameter changes not logged with operator ID and timestamp.
⚙️ Root Cause: Machine PLC operating on GAMP5 Category 3 firmware without 21 CFR Part 11-compliant audit trail module — common in machines manufactured before 2018 that have not undergone software re-qualification.
🔧 Forester's Fix: Upgrade PLC firmware to GAMP5 Category 4 configuration with: (a) immutable audit trail logging operator ID + timestamp for all parameter changes ≥1% of setpoint; (b) role-based access control with ≥3-tier user authorization levels; (c) electronic signature on batch record approval. All HIJ Machinery DPP-series machines ship with 21 CFR Part 11-ready Siemens S7-1200/1500 PLC with pre-configured audit trail — zero post-delivery software modification required. -
🔴 Symptom: OQ sealing temperature mapping shows ≥8°C variance across the sealing jaw width at nominal setpoint — OQ protocol fails acceptance criteria of ±3°C.
⚙️ Root Cause: Uneven resistance heater element degradation within the jaw casting — zones nearest the thermocouple feedback sensor run at setpoint while far-edge zones run 8–12°C cooler after >3 years of service cycles.
🔧 Forester's Fix: Replace full sealing jaw heater assembly (not individual elements) and re-map temperature at 9 grid points after 30-minute thermal stabilization. If variance persists >5°C after heater replacement, the jaw casting itself has developed micro-fractures — replace casting. Document all corrective actions in the OQ deviation log with root cause analysis per EU GMP Annex 15 change control procedures.
How to Select the Right Pharmaceutical Blister Packaging Machine: 7 Critical Factors Every Procurement Engineer Must Evaluate
Selecting a pharmaceutical blister packaging machine requires simultaneous evaluation of 7 interdependent engineering and compliance factors. Optimize for only 2–3 of these factors while ignoring the others, and the system will fail — either mechanically during production ramp-up or documentarily during your first regulatory inspection.

Fig. 7: Seven-factor machine selection framework for pharmaceutical blister packaging procurement — validated by HIJ Machinery across 200+ project deployments in Southeast Asia, Latin America & Middle East.
| # | Selection Factor | Key Questions to Ask the Vendor | Red Flag Answer | HIJ Machinery Answer |
|---|---|---|---|---|
| 1 | Target Output Speed | What is the validated OEE at rated speed with your specific product geometry? | “Maximum speed” without OEE qualification data | OEE ≥85% demonstrated during PQ with actual product |
| 2 | Film Material Compatibility | Has the machine been validated with PVDC/Aclar®/Alu-Alu at your cavity geometry? | “Compatible with all films” without test data | Factory test results with specific film & cavity spec provided before PO |
| 3 | Regulatory Jurisdiction | What validation documentation package (IQ/OQ/PQ) is included? Is it WHO, FDA, or EU GMP formatted? | “We can prepare documentation after delivery” | Full DQ/IQ/OQ templates delivered with machine; PQ support on-site |
| 4 | Change-Part & Tooling System | What is the average changeover time between SKUs? Is it tool-free? | >4 hours changeover; no documented change-part procedure | <45 min changeover via quick-release format parts; full change-part doc supplied |
| 5 | PLC & Electronic Records | Is the PLC audit trail 21 CFR Part 11 compliant? What GAMP5 category? | “We can add that feature” — not pre-configured | Siemens S7-1200/1500 with pre-configured 21 CFR Part 11 audit trail |
| 6 | Downstream Integration | Has the blister machine been integrated with your cartoner model? Are PLC protocols compatible? | “Integration is the cartoner vendor's responsibility” | HIJ Turnkey integration: one contract covers blister + cartoner PLC synchronization |
| 7 | After-Sales & Spare Parts | Where is the nearest service engineer? What is guaranteed spare part lead time? | “Please contact your local agent” — no direct support | HIJ regional engineers in Southeast Asia, Latin America, Middle East; 72-hr spare parts delivery |
Based on HIJ Machinery's commissioning data across 200+ pharmaceutical packaging projects, facilities that procure blister packer + cartoner as a single integrated turnkey system achieve first-pass SAT success in 91% of projects vs. 47% success rate for multi-vendor assembled lines. The 44% gap is almost entirely attributable to PLC integration failures and documentation accountability gaps — not to individual machine quality. Source: HIJ Machinery project database, 2019–2024. See our turnkey pharmaceutical packaging solutions for full integration scope.
Common Failure Modes & Forester's Fix — Machine Selection & Integration Errors
-
🔴 Symptom: Blister packer and cartoner run at rated individual speeds, but integrated line achieves only 65% of target throughput — chronic jam-induced stoppages at the cartoner infeed.
⚙️ Root Cause: Blister card discharge pitch (spacing between cards) from die-cutter is 2–3 mm inconsistent — caused by incorrect blister machine index servo tuning — creating double-card feeding events at the cartoner pusher every 12–20 cycles.
🔧 Forester's Fix: Tune index servo deceleration ramp to 180 ms (from factory default 240 ms) to reduce blister card trailing momentum on discharge conveyor. Set conveyor belt speed 8–12% faster than blister machine discharge rate to create consistent card separation gaps of ≥15 mm. Conduct integrated SAT at 80%, 90%, and 100% rated speed — do not sign SAT acceptance until 30-minute continuous run at 100% speed with <0.5% jam rate. -
🔴 Symptom: After 6 weeks of production, regulatory inspector flags that the blister machine's process parameter logs show 3-minute gaps in temperature recording during shift changeovers.
⚙️ Root Cause: PLC data historian configured to log every 60 seconds by default, but operator “sleep mode” activation during changeovers pauses the historian — a 21 CFR Part 11 non-compliance gap as parameter continuity cannot be assured during the unlogged periods.
🔧 Forester's Fix: Reconfigure PLC historian to continuous logging mode (10-second intervals) with operator lockout preventing historian pause without supervisor-level electronic signature and mandatory deviation justification entry. Archive all historian data to RAID-protected server with 5-year retention per 21 CFR Part 211.68 electronic records policy.
HIJ Machinery Pharmaceutical Blister Packaging Machines: Complete Product Line & Technical Specifications
HIJ Machinery designs and manufactures a complete range of pharmaceutical blister packaging machines — from entry-level semi-automatic units for clinical-trial batch production to high-speed fully automatic GMP blister lines for commercial manufacturing. Every machine ships with CE marking, WHO GMP-compliant structural design, and full IQ/OQ documentation templates — ready for regulatory inspection from day one.




Frequently Asked Questions: Pharmaceutical Blister Packaging — What Buyers & QA Engineers Ask Most
Ready to Engineer Your Pharmaceutical Blister Packaging Line?
Tell Forester your API moisture sensitivity, target market regulatory jurisdiction, and production volume — and receive a technically detailed machine recommendation within 24 hours, not a generic catalog PDF.
— Forester Xiang, Founder & Chief Engineer, HIJ Machinery








