What Is Alu Alu Blister Packaging?
Alu-Alu blister packaging is defined as a pharmaceutical primary packaging format in which both the forming web and the lidding foil are constructed from cold-rolled aluminum laminate — typically OPA/Al/PVC or OPA/Al/PE — creating a hermetically sealed unit-dose cavity with a water vapor transmission rate (WVTR) below 0.5 g/m²/day, near-zero oxygen permeability, and zero light transmission. No thermoforming heat is applied; mechanical cold-pressing preserves 100% foil continuity, which is the physical basis for its near-absolute barrier performance. For hygroscopic APIs, beta-lactam antibiotics, and biologics requiring storage in WHO climate Zone III/IV, cold-form aluminum blister is not a preference — it is a regulatory requirement.
What Is Alu Alu Blister Packaging? The Complete Guide to Cold Form Aluminum Protection

HIJ DPH-300 Cold Form Alu-Alu Blister Packaging Machine — engineered for absolute barrier pharmaceutical packaging of hygroscopic APIs.
How Cold Forming Technology Delivers an Absolute Moisture Barrier
Cold forming technology operates on a fundamentally different physical principle than thermoforming: rather than softening a polymer film with heat and drawing it into a cavity mold, cold forming mechanically presses a multi-layer aluminum laminate at ambient temperature through a precision die, forcing the metal to plastically deform into the pocket shape. The aluminum layer — typically 60–80 µm thick — stretches and work-hardens without any heat-induced thinning, maintaining structural continuity across the entire cavity surface.
This distinction is the entire reason alu-alu blister packaging achieves near-absolute barrier performance. Thermoforming PVC or PVDC film at 120–160°C creates micro-thinning at cavity corners and sidewalls, raising local WVTR values beyond the bulk film specification. Cold-pressed aluminum has no such weakness: the foil deforms without heating, and its crystalline metal lattice — even when thinned to 40–45 µm at the deepest draw points — remains impermeable to water vapor molecules. Per USP <1207> and ASTM F2338 package integrity testing protocols, cold-form aluminum blisters consistently achieve WVTR values below 0.5 g/m²/day at 40°C/75% RH (ICH Q1B Zone IVB accelerated conditions).

Cold forming station on the HIJ DPH-300: mechanical die pressure at room temperature — no heat, no polymer thinning, zero barrier compromise.
Cold Forming vs Thermoforming: The Physics of Barrier Integrity
| Parameter | Cold Forming (Alu-Alu) | Thermoforming (PVC/Alu or PVDC/Alu) |
|---|---|---|
| Forming mechanism | Mechanical die press at ambient temperature | Heat softening + vacuum/pressure draw at 120–160°C |
| Forming web material | OPA/Al/PVC or OPA/Al/PE laminate | PVC, PVDC, PET, or PCTFE film |
| WVTR (40°C/75%RH) | < 0.5 g/m²/day | 0.5 – 3.0 g/m²/day (grade-dependent) |
| Oxygen transmission | ≈ 0 cm³/m²/day | 0.1 – 5.0 cm³/m²/day |
| Light barrier | 100% opaque (zero UV/visible light) | Transparent to UV unless pigmented |
| Max cavity depth | Up to 12 mm (HIJ DPH-300) | Up to 20 mm (PVC unlimited thermal draw) |
| Cavity corner risk | None — metal deforms without thinning failure | Micro-thinning at corners raises local WVTR |
| Pack appearance | Matte silver, opaque both sides | Clear/translucent forming web (product visible) |
| Regulatory target | Class I hygroscopic APIs, Zone IVB markets | Standard moisture-tolerant oral solids |
| Material cost premium | +30–50% vs PVC/Alu | Baseline reference cost |
— Forester Xiang, Founder, HIJ Machinery
Common Failure Modes & Forester's Fix
-
🔴 Symptom: Visible pinholes or micro-cracks on the cold-formed aluminum cavity floor after sealing.
⚙️ Root Cause: OPA/Al/PVC forming foil aluminum gauge too thin (< 45 µm) or forming die clearance set 0.03–0.05 mm too tight, causing foil to fracture under excessive tensile stress at cavity base during cold-press stroke.
🔧 Forester's Fix: Specify OPA/Al/PVC with minimum 60 µm aluminum layer. Set forming die punch-to-cavity clearance at 0.10–0.15 mm per material datasheet. Run peel-force verification per ASTM F88 at ≥ 8 N/15mm before production. -
🔴 Symptom: Sealing bond failure between cold-form web and aluminum lidding foil — blisters peel open under 5 N/15mm pull test.
⚙️ Root Cause: Heat-sealing temperature set below 180°C or dwell time under 0.6 seconds, insufficient for full PVC-to-Alu heat-seal lacquer fusion; also caused by foil roll humidity exposure > 60% RH during changeover.
🔧 Forester's Fix: Set sealing station temperature to 200°C ± 5°C, dwell time 0.8–1.0 seconds, sealing pressure 280–320 kPa. Store foil rolls in sealed moisture-barrier bags; expose to ambient air for < 30 minutes before mounting per EU GMP Annex 1 (2022) Section 8.10 environmental control requirements. -
🔴 Symptom: Cavity depth inconsistency > ± 0.5 mm across a single forming web strip, causing tablets to sit too high and lid sealing to be uneven.
⚙️ Root Cause: Forming die punch guide wear > 0.08 mm lateral play, or foil feed tension inconsistency > ± 5% between forming strokes.
🔧 Forester's Fix: Inspect forming punch guide bushing wear with a 0.01 mm dial gauge every 2 million strokes; replace at > 0.05 mm play. Calibrate servo foil feed tension to ± 2% tolerance; verify cavity depth with CMM measurement during IQ/OQ protocol per EU GMP Annex 15.
What Products Require Alu Alu Blister Packaging — And Why Choosing Wrong Costs You a Market
Alu-Alu blister packaging is the validated primary packaging format for any active pharmaceutical ingredient (API) classified as ICH Q1B Class I moisture-sensitive — defined as products that degrade measurably at ≥ 25°C/60% RH when packaged in standard PVC/Alu blister. The regulatory imperative intensifies for products targeting WHO climate Zone IVB markets (Indonesia, India, Nigeria, Brazil): ambient conditions of 30°C/75% RH sustained year-round make absolute barrier packaging non-negotiable for shelf-life compliance.
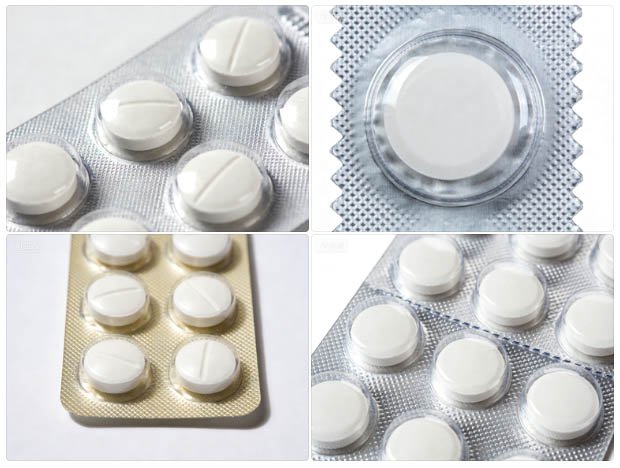
Finished alu-alu blister packs: opaque silver on both sides — every cavity hermetically sealed with zero moisture, oxygen, or light transmission.
📌 Drug Categories That Mandate Cold-Form Aluminum Blister
| Drug Category | Representative APIs | Degradation Mechanism Without Alu-Alu |
|---|---|---|
| Beta-lactam antibiotics | Amoxicillin, Ampicillin, Piperacillin | Beta-lactam ring hydrolysis accelerated by moisture; potency loss > 5% within 6 months in PVC/Alu at 30°C/75% RH |
| Psychiatric / CNS | Clozapine, Lithium carbonate, Olanzapine | Moisture-catalyzed oxidation and crystal form changes alter bioavailability |
| Effervescent tablets | Vitamin C effervescent, Antacid | Premature CO&sub2; release and tablet disintegration at > 0.5 g/m²/day WVTR |
| Hygroscopic biologics | Lyophilized peptide tablets, Enzyme replacement | Protein denaturation and aggregation at > 5% tablet moisture content |
| Oxygen-sensitive generics | Furosemide, Nifedipine | Photodegradation and oxidative ring-opening amplified by UV exposure through transparent PVC |
| Controlled-release formulations | Extended-release metformin, Osmotic tablets | Moisture ingress disrupts polymer matrix swelling kinetics, altering release profile |

In my 20 years auditing pharma lines across Southeast Asia and Latin America, I've watched procurement teams get burned by the same mistake repeatedly: purchasing a standalone cold-form blister machine at a discount, only to discover it lacks validated PLC protocols for line integration and ships with zero cGMP compliance documentation.
The hidden trap is the integration gap. Your $80K machine becomes a $300K problem the moment it fails FAT/SAT or gets flagged during an FDA pre-approval inspection. I've seen a Bangladesh manufacturer lose a full 18-month registration timeline because their "alu-alu capable" machine had no electronic batch record function — a direct 21 CFR Part 11 non-conformance. I've seen an Indonesian plant re-pack 400,000 amoxicillin blisters by hand after their unvalidated cold-form machine produced WVTR failures detected during WHO TRS 992 Annex 3 stability testing.
Before specifying any alu-alu line, lock down your URS first — forming depth, cavity tolerance (± 0.1 mm), sealing temperature range, and downstream cartoning compatibility. At HIJ, our turnkey philosophy means the blister machine, feeder, and cartoning machine are validated as a single system, eliminating multi-vendor finger-pointing and compressing your time-to-market by an average of 4–6 months.
Common Failure Modes & Forester's Fix
-
🔴 Symptom: Stability testing failure at 6-month ICH accelerated conditions (40°C/75% RH) — tablet assay drops > 5% despite apparently intact alu-alu blisters.
⚙️ Root Cause: Forming foil aluminum gauge below specification (< 45 µm actual vs 60 µm spec) due to unverified incoming material; WVTR failure at cavity corners was undetected without in-process leak testing per USP <1207>.
🔧 Forester's Fix: Implement 100% incoming foil thickness measurement (eddy-current gauge, ± 1 µm accuracy). Add blue-dye immersion or vacuum decay leak test per ASTM F2338 at start-up and every 500 cycles in-process. Reject any batch with foil gauge variance > ± 3 µm. -
🔴 Symptom: Regulatory audit finding: no 21 CFR Part 11-compliant electronic batch records for alu-alu line — all parameters recorded on paper logbooks.
⚙️ Root Cause: Machine PLC does not support audit trail functionality; operator can overwrite process parameters without access control or timestamped record.
🔧 Forester's Fix: Specify Siemens S7-1500 PLC with WinCC SCADA integration providing: role-based access (21 CFR Part 11 §11.10(d)), full audit trail with operator ID + timestamp, electronic batch record export to PDF/A. HIJ DPH-300 ships with this configuration as standard; request IQ/OQ protocol package before purchase.
Alu Alu vs PVC Alu Blister Packaging: Full Barrier Performance Comparison
The decision between cold-form alu-alu and thermoformed PVC/Alu (or PVDC/Alu) blister packaging is ultimately a function of three variables: the API's moisture sensitivity class, target market climate zone, and required shelf-life duration. Per WHO TRS 992 Annex 3 stability testing guidelines, products claiming 24-month shelf life at Zone IVB conditions (30°C/75% RH) must demonstrate stability in their proposed primary packaging — and PVC/Alu packaging simply cannot provide the WVTR values required for Class I hygroscopic APIs to pass accelerated stability.

Blister packaging material comparison: PVC vs PVDC vs Alu-Alu forming web — WVTR, OTR, and cost positioned for pharma decision-makers.
| Barrier Parameter | Alu-Alu (OPA/Al/PVC) | PCTFE/Alu (Aclar®) | PVDC/Alu (220 g/m²) | PVDC/Alu (90 g/m²) | PVC/Alu (250 µm) | PVC/Alu (200 µm) |
|---|---|---|---|---|---|---|
| WVTR (g/m²/day) 38°C/90%RH | < 0.05 | 0.07 – 0.15 | 0.20 – 0.40 | 0.80 – 1.20 | 2.00 – 3.00 | 3.00 – 5.00 |
| OTR (cm³/m²/day) | ≈ 0 | < 0.1 | 0.5 – 2.0 | 3.0 – 8.0 | > 10 | > 15 |
| Light barrier | 100% opaque | Transparent | Transparent | Transparent | Transparent | Transparent |
| ICH Zone IVB compliance | ✅ Full | ✅ Full | ⚠️ API-dependent | ✖ Class I fails | ✖ Class I fails | ✖ Class I fails |
| Relative material cost | 3.0× | 4.5× | 2.0× | 1.5× | 1.0× (baseline) | 0.85× |
| Child-resistant option | Yes (peel-push design) | Yes | Yes | Yes | Yes | Limited |
| Recyclability | Aluminum recovery possible | Difficult | Difficult (PVDC halogen) | Difficult | Limited | Limited |
Compliance note: Under 21 CFR Part 211.94, drug product containers and closures must be shown to be suitable for their intended use, with data demonstrating they protect the drug product from moisture, light, and other environmental factors that could cause degradation. For hygroscopic APIs, regulatory agencies expect submission data generated from the actual primary packaging used in commercial production — switching from alu-alu to PVC/Alu post-approval requires a Prior Approval Supplement. EU GMP Annex 1 (2022) Section 8.10 further mandates documented environmental controls during blister packaging of sterile and non-sterile products with absolute barrier specifications.
Common Failure Modes & Forester's Fix
-
🔴 Symptom: WVTR results from third-party lab show 1.2 g/m²/day on finished alu-alu blisters — far above the < 0.5 target — despite using specification-grade OPA/Al/PVC forming foil.
⚙️ Root Cause: Sealing station temperature uniformity across 320 mm sealing die width was ± 18°C (acceptable limit: ± 5°C), leaving cold zones where heat-seal lacquer did not achieve full fusion — creating a 0.3–0.8 mm unsealed perimeter around each cavity.
🔧 Forester's Fix: Map sealing die temperature with 16-point thermocouple array before production (IQ protocol requirement per EU GMP Annex 15). Replace sealing die heater cartridges if temperature uniformity > ± 5°C. Target sealing parameters: 200°C ± 3°C, 300 kPa ± 10 kPa, dwell 0.9 seconds. -
🔴 Symptom: Regulatory submission rejected: stability data generated on PVC/Alu packaging, but commercial production intends alu-alu — agency issues deficiency letter requiring bridging stability study.
⚙️ Root Cause: Packaging material specification in CTD Module 3.2.P.7 did not match IND/NDA proposed commercial primary packaging, triggering 21 CFR §314.70 Prior Approval Supplement requirement.
🔧 Forester's Fix: Lock primary packaging specification (foil grade, gauge, WVTR value) in URS before IND submission. Generate initial stability data in commercial alu-alu packaging at ICH Q1A accelerated conditions (40°C/75% RH, 6 months minimum) to support NDA/MAA Module 3.2.P.8.3.
How HIJ's Alu Alu Blister Packing Machine Delivers Validated Absolute Barrier Protection
HIJ Machinery's alu alu blister packing machine — the DPH-300 series — is engineered specifically for cold-form aluminum pharmaceutical packaging, combining precision servo-driven forming mechanics with a full EU GMP Annex 1 and 21 CFR Part 11-ready control architecture. Unlike entry-level cold-form machines that bolt on a forming station to a thermoforming frame, the DPH-300 is built from the ground up as a cold-forming system: the forming station, feeding system, sealing station, and die-cutting unit are all sized and calibrated for the mechanical forces of aluminum laminate processing, not polymer film.

HIJ DPH-300 Cold Form Alu-Alu Blister Packing Machine
The DPH-300 delivers absolute barrier cold-form blister packaging for hygroscopic APIs, beta-lactam antibiotics, and moisture-sensitive tablets — with full FAT/SAT validation support and 21 CFR Part 11-compliant electronic batch records as standard.
- Output speed: Up to 300 blisters/min
- Max forming depth: 12 mm (cold-press, aluminum laminate)
- Forming foil width: Max 320 mm
- Servo accuracy: ± 0.1 mm cavity positioning
- Sealing temperature: 160–220°C ± 3°C (PID control)
- PLC platform: Siemens S7-1500 + WinCC SCADA
- Compliance: EU GMP Annex 1, 21 CFR Part 11, CE
- Validation docs: IQ/OQ/PQ protocols standard

DPH-300 sealing station: dual-zone PID temperature control with ± 3°C uniformity across the full 320 mm die width — validated to EU GMP Annex 15 IQ/OQ protocols.
The HIJ Turnkey Integration Advantage
Procuring a cold-form blister machine in isolation is one of the most expensive decisions a pharma plant manager can make — not because of the machine price, but because of the validation cost incurred when upstream feeders and downstream blister cartoning machines are sourced from different vendors with incompatible communication protocols. HIJ's turnkey packaging line integration approach validates the complete system — from feeder through blister machine through cartoner — as a single functional unit under one DQ/IQ/OQ/PQ validation package. This approach eliminates multi-vendor finger-pointing and compresses average time-to-GMP-readiness by 4–6 months versus piecemeal procurement.
-
URS Definition & Forming Depth Verification — Forester's team reviews your API moisture sensitivity class, cavity dimensions (length × width × depth ± 0.1 mm), and required output speed to size the DPH-300 configuration before any commercial commitment.
-
Tooling & Mold Engineering — Cold-form punch-and-die tooling is precision-machined to ± 0.02 mm tolerance for your cavity geometry; mold steel hardness HRC 58–62 for 50+ million cycle service life.
-
FAT (Factory Acceptance Test) — Full DQ/IQ/OQ executed at HIJ's facility in Xinyang; sealing integrity validated per ASTM F2338 blue-dye immersion; electronic batch record functionality demonstrated per 21 CFR Part 11 §11.10.
-
SAT (Site Acceptance Test) & OQ at Customer Facility — HIJ engineers install, calibrate, and re-validate on-site; OQ challenge runs at ± 10% of nominal speed with statistical batch record review.
-
PQ & First Production Release — Process Qualification with three consecutive production batches; WVTR verification on finished blisters; IQ/OQ/PQ documentation package delivered in CTD-ready format.

HIJ engineer conducting on-site SAT and OQ validation for an alu-alu blister line installation — Southeast Asian pharmaceutical client, cleanroom ISO Class 8 environment.
Common Failure Modes & Forester's Fix
-
🔴 Symptom: DPH-300 integration with third-party cartoner fails during SAT — blister transfer conveyor speed mismatch causes 12–15 blister pile-up jams per hour.
⚙️ Root Cause: Cartoner infeed speed not synchronized to blister machine output via shared PLC handshake; two independent servo controllers operating on different timing cycles.
🔧 Forester's Fix: Implement Profinet IO master/slave synchronization between DPH-300 Siemens S7-1500 PLC and cartoner PLC; set blister transfer conveyor speed to 105% of blister machine nominal cycle rate as buffer. This is standard in HIJ turnkey line integration but must be custom-engineered for third-party cartoners. -
🔴 Symptom: FDA pre-approval inspection finds that DPH-300 sealing temperature data cannot be extracted from PLC for batch record — inspector issues 483 observation for 21 CFR Part 211.68 deficiency.
⚙️ Root Cause: Machine purchased without WinCC SCADA option; PLC stores only last 100 temperature readings in volatile RAM; no WORM-compliant data storage configured per 21 CFR Part 11 §11.10(c).
🔧 Forester's Fix: Specify DPH-300 with WinCC V16+ SCADA and SQL database logging to WORM-enabled NAS storage (≥ 7-year retention). HIJ provides 21 CFR Part 11 compliance matrix document mapping all 36 Part 11 requirements to specific DPH-300 software functions — request this document during RFQ.
Building a Complete Alu Alu Blister Packaging Line: Upstream and Downstream Integration
Alu-alu blister packaging performance is only as strong as the weakest integration point in the surrounding production line. Cold-form blister machines operate at up to 300 blisters/min, requiring precision-timed feeding of tablets or capsules into cavities — any interruption propagates as empty-cavity rejects and batch record exceptions under cGMP. HIJ's complete pharmaceutical blister packing machine ecosystem covers every upstream and downstream integration point.
| Line Position | HIJ Equipment | Integration Interface with DPH-300 |
|---|---|---|
| Capsule filling (upstream) | Automatic Capsule Filling Machine | Discharge conveyor with speed-synchronized servo handshake |
| Tablet/capsule feeding | Vibrating brush feeder (integrated in DPH-300) | Direct mount; cavity fill rate verified by camera vision system |
| Blister cartoning | Blister Cartoning Machine | Profinet IO synchronization; blister count and leaflet insertion verified |
| Secondary packaging | Case Packing Machine | Carton count accumulation conveyor; barcode traceability handshake |

A complete HIJ pharmaceutical blister packing line: capsule filler → cold-form alu-alu blister machine → blister cartoner → case packer, validated as a single integrated system.
Common Failure Modes & Forester's Fix
-
🔴 Symptom: Empty cavity rate exceeds 0.5% AQL limit during alu-alu blister production of hygroscopic capsules — batch rejected during in-process quality check.
⚙️ Root Cause: Vibrating brush feeder amplitude set for tablet density (1.2 g/cm³) but capsules have density 0.8 g/cm³; capsules bouncing out of cavities before lidding foil sealed.
🔧 Forester's Fix: Reduce brush feeder vibration amplitude by 30–40% for capsule products; add downstream camera inspection system (resolution 0.2 mm per pixel) with automatic reject gate ejecting empty-cavity blisters before sealing station. Set camera detection sensitivity threshold at 15% cavity fill level. -
🔴 Symptom: Cartoner leaflet insertion failure rate of 3% during high-speed operation (> 250 blisters/min), causing batch rejection at secondary packaging QC.
⚙️ Root Cause: Blister transfer timing between DPH-300 and blister cartoning machine off by 0.08 seconds at high speed, causing blister to arrive at cartoner pusher station 15 mm out of position.
🔧 Forester's Fix: Re-tune Profinet IO synchronization master clock offset; add encoder feedback on transfer conveyor belt; verify ≤ 0.5 mm positional accuracy at 250 blisters/min using stroboscope alignment check. This is standard HIJ after-sales service commissioning step.
Frequently Asked Questions About Alu Alu Blister Packaging
-
What is alu alu blister packaging?
Alu-Alu blister packaging is defined as a pharmaceutical primary packaging format in which both the forming web and the lidding foil are constructed from cold-rolled aluminum laminate — typically OPA/Al/PVC (oriented polyamide / aluminum / PVC) or OPA/Al/PE — creating a hermetically sealed unit-dose cavity with WVTR below 0.5 g/m²/day and zero light transmission. Unlike PVC/Alu blister where only the lidding is aluminum, alu-alu blisters have aluminum on both top and bottom, achieving near-absolute moisture barrier performance classified as the highest protection level in pharmaceutical primary packaging. For more on the machine technology that produces this format, see HIJ's alu alu blister packing machine specifications.
-
What is the difference between alu alu and PVC alu blister packaging?
The fundamental difference between alu-alu and PVC/Alu blister packaging is the forming web material: alu-alu uses cold-pressed aluminum laminate (WVTR < 0.5 g/m²/day), while PVC/Alu uses thermoformed polymer film (WVTR 2.0–5.0 g/m²/day for standard PVC grades). This 10–20× difference in moisture barrier performance determines regulatory compliance for hygroscopic APIs in tropical climate markets. Alu-alu is also fully opaque (100% light barrier), while PVC/Alu is transparent on the forming side. The trade-off: alu-alu forming foil costs approximately 3× more per square meter than standard PVC film, and cold-form cavities are limited to approximately 12 mm depth versus unlimited thermoform draw depth.
-
Which drugs require alu alu cold form blister packaging?
Drugs that require alu-alu cold form blister packaging include: beta-lactam antibiotics (amoxicillin, ampicillin, piperacillin), psychiatric medications (clozapine, lithium carbonate, olanzapine), effervescent tablets (vitamin C, antacids), lyophilized peptide tablets, and any API classified as ICH Q1B Class I moisture-sensitive. WHO TRS 992 Annex 3 mandates absolute barrier primary packaging for products targeting Zone IVB markets (30°C/75% RH) with claimed shelf life ≥ 24 months. The practical test: if your API shows > 5% assay degradation in PVC/Alu packaging at 40°C/75% RH accelerated stability conditions within 6 months, alu-alu is required.
-
How much does an alu alu blister packing machine cost?
An alu-alu blister packing machine costs between $60,000 and $350,000 USD depending on output speed, forming web width, PLC/SCADA configuration, and included validation documentation. Entry-level cold-form machines producing 60–120 blisters/min typically range $60,000–$120,000; mid-range machines like the HIJ DPH-300 at 300 blisters/min with full 21 CFR Part 11 compliance and IQ/OQ/PQ documentation range $150,000–$220,000; high-speed pharmaceutical lines > 400 blisters/min can exceed $350,000. The critical cost consideration is not machine price but total validation cost: a machine without cGMP documentation can cost $200,000–$400,000 more in remediation versus one that ships with a complete validation package. Contact HIJ Machinery for a project-specific quotation.
Get Forester's Expert Guidance — From URS to GMP-Ready Production
HIJ Machinery delivers cold-form alu-alu blister packaging lines with complete IQ/OQ/PQ validation packages, 21 CFR Part 11 & EU GMP Annex 1 compliance documentation, and integrated cartoning — serving pharmaceutical manufacturers across Southeast Asia, Latin America, the Middle East, and Africa. Tell us your API, forming depth, and target output; Forester's team responds within 24 hours with a project-specific proposal.







