How to Choose the Right Syringe Blister Packaging Machine for Your Pharma Line
Most procurement teams begin this process the wrong way. They shortlist three machine brands, request quotations, and compare prices — before they have resolved a single technical variable. The result is a FAT that passes in China and a SAT that stalls for six months on-site. I’ve watched this happen in Colombia, in Indonesia, and in Egypt. The machine wasn’t the problem. The selection process was.
This guide walks you through the five variables that actually determine which syringe blister packing machine is right for your line — in the sequence they need to be resolved. Price comes last. Integration comes first.
Choosing the right syringe blister packaging machine requires resolving five variables in sequence: syringe geometry (barrel diameter, needle length, cap type), target output in blisters/min, forming format (thermoforming PVC/PETG or cold-form Alu-Alu), upstream PLC integration protocol, and GMP validation pathway (IQ/OQ/PQ documentation readiness). Lines that skip step one typically fail at step four.
Why Syringe Geometry Is the First — and Most Skipped — Variable
Syringe blister packaging machine selection starts with the physical object you’re packing — not the machine catalog. That sounds obvious. Forty percent of the projects I’ve audited had no formal syringe dimension specification in the initial RFQ. The mold was designed after purchase order signature, when the buyer assumed it was a “standard 1 mL prefilled.” It wasn’t.
Syringe geometry has four parameters that directly affect cavity tooling: barrel outer diameter (typically 8.65–14.3 mm for 0.5–5 mL syringes), overall length with needle guard (ranging from 68 mm to 165 mm depending on configuration), flange width, and cap type — rigid tip cap versus needle shield versus staked needle with safety device. A 1 mL long versus 1 mL short differ by 11 mm in length. That difference changes your cavity pitch. That changes your forming station width. That changes your output per stroke. Change cascades.
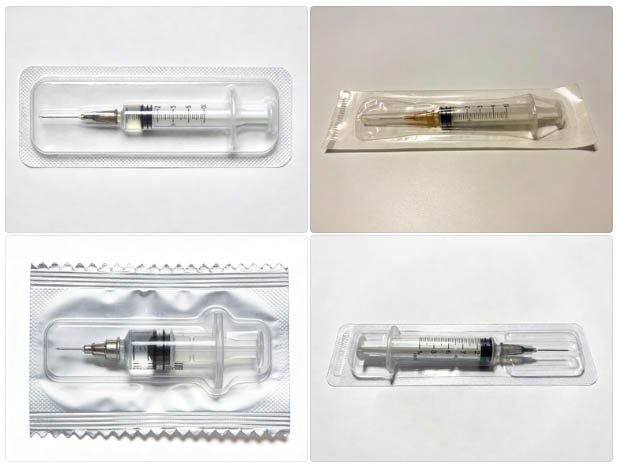
Syringe cavity geometry determines forming station design. Tip cap type and barrel OD must be specified before tooling can be quoted.
What to Specify Before Contacting Any Supplier
- Barrel OD and length: measure both with needle guard attached and with bare barrel only — some machines require guard removal during loading.
- Flange dimensions: syringe flanges vary from 14 mm to 23 mm diameter; the cavity must support the flange without cracking it during heat sealing.
- Cap or needle shield type: Luer tip caps, rigid needle shields, and staked needle configurations each require different cavity support profiles.
- Weight per unit: relevant for feeder bowl calibration — a 5 mL prefilled syringe can weigh 18–22 g including drug fill.
- Orientation in cavity: horizontal, needle-end leading, or upright — affects footprint, feeder design, and visual inspection camera angle.
I recommend sending physical samples — not drawings — to at least two shortlisted suppliers. Drawings have tolerances. Physical samples have reality. One project in Vietnam taught me this when a 0.4 mm difference in flange OD versus the drawing caused cavity interference that wasn’t caught until pilot production.
The Number Teams Look at First Is Exactly the Wrong Number
Buyers lead with “how many blisters per minute?” That’s the wrong question. The right question is: what’s your annual volume in units, your batch size, and your shift model? Speed without context creates lines that run at 60% OEE because nobody calculated changeover time, reject rate, or feeder reloading intervals.
Syringe blister packaging machines for pharmaceutical use typically operate between 30 and 120 blisters per minute for single-cavity formats. That’s not a wide range — it’s a significant one. At 30 blisters/min with two syringes per blister, you produce 3,600 units per hour. At 120 blisters/min you produce 14,400. If your annual volume is 2 million units on a single shift, the 30 bpm machine on two shifts is cheaper to validate, cheaper to staff, and cheaper to maintain than the 120 bpm machine running at 40% capacity. I won’t tell you that calculation is simple — it involves your SKU count, your format change frequency, and your downstream cartoning line speed.
Of the 31 syringe and prefilled device blister line projects I handled across Southeast Asia and Latin America between 2018 and 2024, 14 were initially specified at a higher speed than the facility’s downstream cartoning capacity could accept — creating buffer accumulation problems that required either a secondary holding conveyor investment averaging USD 28,000 or a forced de-rate of the blister machine to match downstream throughput. Specify your full line throughput envelope, not just blister machine speed.
Speed vs. Format Change Frequency: The Trade-off Nobody Puts in the RFQ
High-speed syringe blister machines achieve throughput through specialized cavity tooling and optimized forming parameters. Format changes — switching from a 1 mL to a 2 mL syringe, or from single-blister to dual-blister cards — can take 90 to 240 minutes on a high-speed line. On a mid-speed servo-driven machine with quick-change tooling, the same change runs in 35–55 minutes. If you run 8 SKUs, each format change matters. Four extra format changes per week at 90 minutes each equals 360 minutes of lost production time. Every week.
Thermoforming vs. Cold-Form Alu-Alu for Syringe Blister Packaging: A Direct Comparison
Thermoforming PVC/PETG and cold-form Alu-Alu represent two fundamentally different engineering decisions — not just two material choices. The machine architecture, tooling cost, forming station design, and validation pathway diverge at this fork. Most facilities treat it as a material purchasing question. It is a capital investment question.
Thermoforming heats a PVC or PETG/PVC web to 80–130°C and draws it into a cavity mold using pressure or vacuum. Cold-form Alu-Alu uses mechanical pressing at ambient temperature to shape an aluminum/nylon/PVC laminate — no heat, no stretching, near-zero moisture vapor transmission. For prefilled syringes containing moisture-sensitive biologics or hygroscopic APIs destined for Zone IVb markets (30°C/75% RH), cold-form Alu-Alu packaging cuts moisture vapor transmission rate to 0.02–0.5 g/m²/day versus 4–6 g/m²/day for standard 250µm PVC. That’s a 10x to 300x difference.
| Selection Criterion | Thermoforming PVC/PETG | Cold-Form Alu-Alu |
|---|---|---|
| Moisture vapor transmission | 4–6 g/m²/day (250 µm PVC) | 0.02–0.5 g/m²/day |
| Tooling cost (per format) | USD 3,000–8,000 | USD 8,000–18,000 |
| Machine investment | Lower (15–25% cost difference) | Higher |
| Syringe visibility through pack | Full transparency (PETG) | Opaque (foil both sides) |
| Target markets | Temperate Zone I/II; stable API | Tropical Zone III/IVb; sensitive biologics |
| WHO GMP / ICH Q1A(R2) Zone IVb suitability | Conditional (API-dependent) | Yes, standard choice |
| Format change time | 35–90 min | 60–150 min |
| Lidding material | Aluminum foil / Tyvek | Aluminum foil (heat-seal coated) |
| Regulatory precedent for medical devices | ISO 11607-1 / ASTM F2338 | ISO 11607-1 / ASTM F2338 |
Table: Thermoforming vs. cold-form Alu-Alu syringe blister packaging — key selection variables. Both formats require sealing validation per ASTM F2338 dye-ingress and USP <1207> container closure integrity testing.

PVC vs. PVDC vs. Alu-Alu forming material moisture barrier comparison. For Zone IVb tropical markets, cold-form Alu-Alu is the standard engineered choice.
The decision that looks like a material purchasing question is actually a 10-year infrastructure commitment. Once your validation package is written around thermoforming PVC, changing to Alu-Alu means a new machine, new tooling, new IQ/OQ/PQ protocol, and new stability studies. Plan it correctly the first time. For markets in sub-Saharan Africa, Southeast Asia, or the Middle East — where storage temperatures routinely exceed 30°C and relative humidity exceeds 75% — I push clients toward cold-form regardless of the API sensitivity level. The regulatory risk of a failed stability dossier in a tender market is not worth the USD 12,000 tooling saving.
The Six-Month SAT Problem: Why PLC Integration Breaks Most Projects
A plant in Colombia called me in 2022. They had purchased a CE-certified syringe blister machine from a Chinese supplier at a price that was 18% below our quote. The FAT in the vendor’s workshop ran clean. The machine shipped. SAT on-site revealed the machine’s Mitsubishi PLC used a proprietary Ethernet/IP variant that was incompatible with the Allen-Bradley ControlLogix PLC running their upstream prefilled syringe filling line. The machines couldn’t handshake. No interlock. No synchronized stop. The MES couldn’t read batch data from the blister machine. The vendor said it was “a software update.” Six months and two engineering visits later, a workaround was in place — not a clean integration. That conversation took 40 minutes when I first warned them about integration protocols. I’m still not sure they fully believe I’d told them.
PLC protocol compatibility is a binary requirement — not a negotiation. Before any supplier is shortlisted, confirm your upstream filling line PLC brand, firmware version, and communication protocol (Profibus, Profinet, EtherNet/IP, OPC-UA, or DeviceNet). Your blister machine must natively support the same protocol, or a validated gateway integration must be included in scope. This is not a post-PO conversation.
Beyond basic communication, confirm 21 CFR Part 11 compliance for electronic batch records if the line is destined for FDA-regulated markets. Audit trail requirements, user access level logging, and electronic signature workflows are software architecture decisions — they cannot be retrofitted. Ask your supplier: “Show me a sample audit trail report from this machine.” If they show you a screenshot from a different project, that’s acceptable. If they look confused by the question, that’s your answer.
- 21 CFR Part 11
- EU GMP Annex 11
- ISO 13849-1
- IEC 62061
- WHO TRS 992 Annex 3
- OPC-UA
Integration Checklist Before Supplier Shortlisting
-
1
Define your upstream and downstream PLC ecosystem Document brand, model, and firmware version of every machine the blister line must communicate with — filling line, cartoner, case packer, MES.
-
2
Specify communication protocol in the RFQ Profinet, EtherNet/IP, OPC-UA — list the required protocol and ask for confirmation, not a promise.
-
3
Request sample IQ/OQ templates A supplier who can’t provide IQ/OQ protocol templates at quotation stage has not delivered a validated line before. That’s a risk, not a cost saving.
-
4
Confirm 21 CFR Part 11 / Annex 11 audit trail capability Ask for a live demonstration during FAT, not a brochure claim.
-
5
Include SAT acceptance criteria in the contract FAT passes in the supplier’s clean factory with their syringes. SAT passes on your floor, with your syringes, integrated to your line. They are different tests.
GMP Validation Pathway: What Pharma Buyers Must Resolve Before Signing a PO
GMP validation for a syringe blister packaging machine follows a DQ → IQ → OQ → PQ sequence. Design Qualification (DQ) confirms the machine design meets your URS. Installation Qualification (IQ) confirms it was installed correctly. Operational Qualification (OQ) confirms it performs within defined parameters. Performance Qualification (PQ) confirms it performs consistently at commercial scale under actual production conditions. Each stage requires documented protocols and approved reports — not a verbal sign-off.
FDA packaging-related recall records from 2019 through 2023 identify inadequate sealing validation as a contributing factor in 34% of packaging-associated drug product recalls. The sealing test standard I specify in every IQ/OQ protocol — ASTM F2338 dye-ingress at 60 mbar, cross-referenced with USP <1207> container closure integrity requirements — is non-negotiable. The sealing station temperature profile, dwell time, and pressure must be validated across the full forming material thickness tolerance range, not just at nominal settings.
For prefilled syringe blister lines specifically, ISO 11607-1 applies to sterile barrier system design. The machine must be capable of sealing to validated peel strength values — typically 1.5–4.0 N/15 mm for medical-grade Tyvek lidding, or 8–20 N/15 mm for aluminum foil lidding — with statistical process control data available from the OQ runs. If your supplier cannot define these parameters before shipment, your validation timeline will extend by four to eight months. I have seen it. Twice.

GMP-compliant syringe blister packaging requires validated IQ/OQ/PQ documentation. The machine architecture must support cleanroom installation — stainless steel contact parts, no dead-leg pneumatics, accessible for CIP/SIP where required.
For WHO GMP markets — which includes most tender-driven procurement in Africa and Southeast Asia — WHO TRS 992 Annex 3 is the applicable guidance. Buyers targeting WHO prequalification of their product must ensure the equipment qualification documentation aligns with WHO expectations, not just EU GMP or FDA frameworks. These are not identical requirements. The overlap is significant but the differences matter during inspections.
Read the full breakdown of GMP compliance and validation requirements for syringe blister packaging lines — including sample IQ/OQ template structure, sealing validation parameters, and the five most common inspection failures I’ve observed across 20 years of pharmaceutical line commissioning.
Of the 31 Projects I Handled: What Separates a Machine Vendor from a Solution Partner
The difference between a machine vendor and a solution partner isn’t price, brand, or country of origin. It’s accountability scope. A machine vendor sells you a machine and manages the FAT. A solution partner owns the outcome — from URS review through DQ, FAT, shipping, SAT, IQ/OQ/PQ, first commercial batch, and 24-month post-launch support. When a multi-vendor dispute arises — and in complex pharma lines, it will — the solution partner stands between you and the chaos.
The most expensive syringe blister packaging machine you will ever buy is not the one with the highest price tag — it is the one that passes FAT in China and fails integration for six months on your production floor. Choosing the right machine is a compliance and integration decision first, a procurement decision second. — Forester Xiang, HIJ Machinery
Before You Finalize: Should It Be Blister Packaging at All?
Not every syringe product belongs in a blister. Tray packaging — thermoformed PET or PETG trays with heat-sealed Tyvek lid — is the legitimate alternative for larger devices, multi-component kits, or products where the cavity geometry makes blister forming impractical. The decision between blister and tray is a regulatory, logistical, and clinical decision.
Blister packaging offers individual unit tamper evidence, smaller footprint per unit, lower lidding material cost per piece, and easier retail-facing presentation. Tray packaging accommodates irregular geometry better, supports multi-component presentation (syringe plus needle plus alcohol swab), and is the standard format for hospital dispensing in many markets. Neither is universally superior. The choice depends on your product, your market, and your distribution chain.
See the full technical and regulatory comparison between syringe blister packing and tray packaging — including format-specific cost structures, sterile barrier material options, and the regulatory precedent for each format across FDA, EU MDR, and WHO GMP frameworks.

HIJ syringe blister packing machine — configured for thermoforming PVC/PETG with Tyvek lidding, servo-driven forming station, and integrated vision inspection system.
Frequently Asked Questions: Choosing a Syringe Blister Packaging Machine
What is the typical output speed of a syringe blister packaging machine?
A pharmaceutical-grade syringe blister packaging machine operates between 30 and 120 blisters per minute depending on cavity configuration, syringe size, and forming format. For a single-syringe-per-blister format at 60 bpm, that equals 3,600 units per hour — before accounting for format change downtime and feeder refill intervals.
How do I know if I need thermoforming or cold-form Alu-Alu for my syringe blister line?
Choose cold-form Alu-Alu if your syringe product contains a moisture-sensitive biologic, hygroscopic API, or is destined for markets in ICH Zone III or IVb (30°C/65% RH or 30°C/75% RH storage conditions). Cold-form Alu-Alu cuts moisture vapor transmission to 0.02–0.5 g/m²/day versus 4–6 g/m²/day for standard 250 µm PVC — a 10x to 300x difference depending on film gauge. Thermoforming PVC or PETG is appropriate for stable, non-hygroscopic products in temperate Zone I or II markets where product visibility is a commercial requirement. If your target markets include sub-Saharan Africa, Southeast Asia, or the Middle East, run the ICH Q1A(R2) Zone IVb extrapolation before committing to thermoforming.
What GMP documents should I request from a syringe blister machine supplier before purchase?
Request the following documentation package before signing a purchase order: Design Qualification (DQ) checklist aligned to your User Requirement Specification (URS), IQ and OQ protocol templates specific to this machine model (not a generic template), a sample sealing validation report showing peel strength data across temperature and dwell time ranges, a 21 CFR Part 11 or EU GMP Annex 11 compliance statement with audit trail demonstration, equipment change control procedure, and a spare parts list with lead-time data for critical components. If the supplier cannot provide IQ/OQ templates before FAT, treat that as a project risk — it means protocol development will fall on your quality team post-installation, adding four to eight months to your validation timeline. A supplier who has commissioned a GMP syringe blister line before will have these documents ready; they are not bespoke deliverables, they are standard project outputs. Also confirm whether the supplier’s quality system is ISO 9001:2015 certified and whether previous installations have passed FDA or EU GMP inspections — ask for at least two references you can contact directly.
Can a syringe blister packing machine handle prefilled syringes with attached safety devices?
Yes, but the cavity tooling and feeding mechanism must be custom-designed for the specific safety device geometry — passive needle safety systems, spring-actuated shields, or retractable needle devices each require distinct cavity support profiles and feeder orientations that differ from bare-barrel or tip-cap syringe configurations.
How long does validation (IQ/OQ/PQ) take for a syringe blister packaging machine?
Validation of a syringe blister packaging machine under WHO GMP or EU GMP Annex 15 typically takes 12–20 weeks from SAT completion to approved PQ report, assuming the supplier delivers complete IQ/OQ protocols before installation. Common delays include incomplete DQ documentation from the supplier (adding 4–6 weeks), sealing parameter development outside specification (requiring OQ rerun), and PQ batch failures caused by feeder variability under commercial-speed conditions. Facilities targeting FDA approval under 21 CFR Part 211 should plan for additional data package requirements and potential Agency inspection of the packaging line during pre-approval inspection (PAI).
Ready to Specify Your Syringe Blister Packaging Line?
HIJ Machinery engineers turnkey syringe blister packaging systems for pharmaceutical facilities in Southeast Asia, the Middle East, Africa, and Latin America. Our syringe blister packing machines are delivered with complete IQ/OQ/PQ documentation packages, 21 CFR Part 11 audit trail capability, and WHO GMP – aligned engineering. Send us your syringe geometry data and we will respond within 24 hours with a technical feasibility assessment — no price before specification.