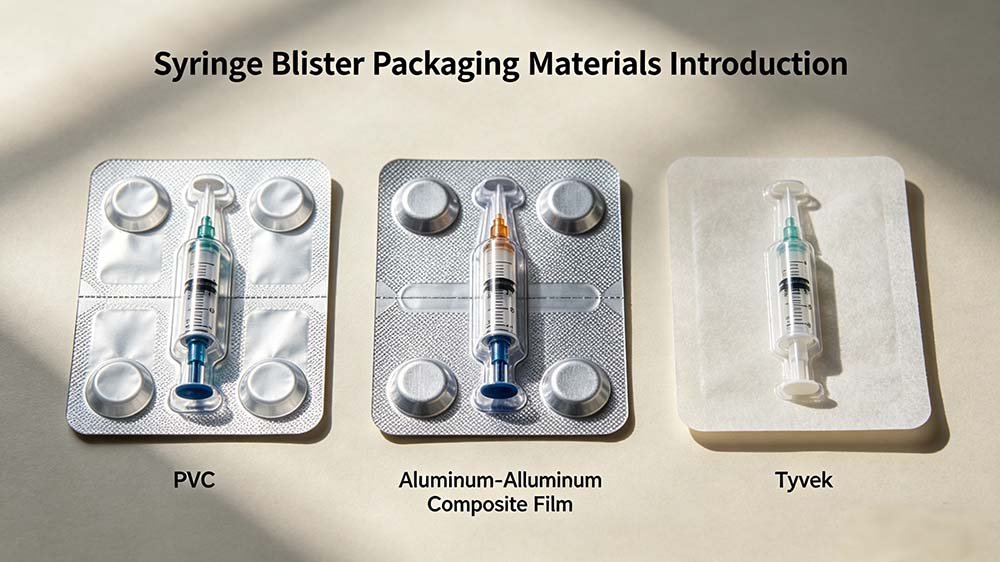
Syringe blister packing thermoforms a film cavity around each individual syringe unit and seals it with a lidding layer — creating a discrete hermetic sterile barrier per unit. Tray packaging thermoforms a rigid multi-cavity tray that holds one or more syringes, sealed with a peelable Tyvek or foil lid — the sterile barrier covers the full tray, not individual units.
Of the 31 syringe packaging line projects I handled in Southeast Asia and Latin America between 2018 and 2023, 19 were blister lines and 12 were tray lines. The split was not random. It tracked almost perfectly with downstream sterilization method and end-market distribution channel.
Blister packaging wraps each syringe in its own sealed cavity — thermoformed PVC/PE or Alu-Alu forming film on the bottom, Tyvek or aluminium lidding on top. Break the seal on one unit, and the other 11 in the carton are untouched. That is the critical advantage in clinic-level distribution where single-unit dispensing is standard.
Tray packaging uses a rigid preformed or thermoformed tray. One lid seals the whole tray. That lid is easier to peel in a sterile field — surgical teams prefer it. But it means one seal failure compromises all units in the tray. That trade-off is the first variable your packaging engineer should map before a machine is ever specified.
EO sterilization requires controlled gas penetration through packaging materials — the cavity depth, material porosity, and seal geometry must be pre-validated with your sterilization contractor before machine specifications are finalised. A blister cavity too deep or too shallow will fail dose mapping and require re-validation at a cost of $15,000–$40,000 per cycle.
The number teams look at first is unit cost per pack. That is exactly the wrong number to start with. I have watched procurement teams in Jakarta and Cairo spend three months comparing machine quotes before anyone called the EO contractor.
Here is what the EO contractor will tell you: they have a validated sterilization cycle for a specific product geometry. That cycle specifies gas concentration, dwell time, humidity, and — critically — the dimensional envelope of the packaging cavity through which EO must diffuse. Change the cavity depth by 4 mm and you are outside validated parameters. You revalidate. You pay again. You delay.
For blister packing, the forming depth for a standard 1 mL prefilled syringe typically runs 18–26 mm. For tray packing of the same syringe, the cavity is shallower — often 12–16 mm — because the tray provides lateral support that blister forming film cannot. That 6–10 mm difference changes your EO dose mapping entirely. It also affects how you design your mold tooling — a capital cost item your machine vendor quotes separately.
Blister packaging achieves per-unit hermetic seal integrity validated to ASTM F2338 at 60 mbar — but only when the cavity geometry matches the EO sterilization contractor’s validated dimensional envelope. Get that alignment before the URS is signed, not after the machine is commissioned. — Forester Xiang, HIJ Machinery
One Brazilian client I worked with in 2022 had locked blister as their format. The EO contractor — a third-party service provider — had validated a tray geometry for a similar product two years earlier and offered a discounted cycle rate if the new product used the same packaging format. The procurement team found out 14 weeks into tooling design. Eleven weeks and $180,000 later, they had a tray line. The machine vendor was blameless. Nobody had asked the right question early enough.
| Decision Variable | Blister Packing | Tray Packaging | Recommendation |
|---|---|---|---|
| Sterile Barrier Standard | ISO 11607 per-unit hermetic seal | ISO 11607 per-tray sterile barrier | Both valid; depends on distribution unit of use |
| EO Gas Penetration | Via Tyvek or porous lidding; cavity depth 18–26 mm must be validated | Easier uniform gas flow over shallow tray; cavity 12–16 mm | Confirm with EO contractor before tooling design |
| Tamper Evidence | Per-unit seal visible on opening; strong retail tamper evidence | Single lid over multiple units; less per-unit evidence | Blister preferred for OTC / clinic distribution |
| Dispensing Environment | Individual unit peel — ideal for single-use clinic setting | Full tray peel — preferred in surgical sterile field | Map end-user workflow before format selection |
| Machine Speed (typical) | 60–150 blisters/min depending on syringe size | 20–60 trays/min (multi-unit per tray) | Output parity depends on units/tray configuration |
| Material Cost per Unit | PVC/PE forming film: $0.008–$0.018/unit; Tyvek lidding: $0.012–$0.025/unit | PETG tray: $0.035–$0.065/tray (multi-unit amortised lower) | Unit-level cost advantage depends on units/tray |
| Mold/Tooling Capex | $8,000–$22,000 per cavity format | $12,000–$30,000 for rigid tray tooling | Blister tooling typically lower per format change |
| Cold Chain / Humidity Sensitivity | Alu-Alu blister achieves 0.02–0.5 g/m²/day MVTR — excellent for Zone IVb | PETG tray with Tyvek lid: 2–5 g/m²/day MVTR — limited moisture barrier | Blister preferred for humid-climate markets (SE Asia, West Africa) |
| GMP Validation Complexity | IQ/OQ/PQ per blister machine; seal integrity per ASTM F2338 | IQ/OQ/PQ per tray line; additional EO cycle validation | Comparable complexity; blister documentation more standardised |
| Regulatory Acceptance | Widely accepted: WHO, FDA, CDSCO, ANVISA, SFDA | Widely accepted: WHO, FDA; check national device regulations | Verify with target market regulator before dossier preparation |
| Line Footprint | Compact: 4–8 m linear for full blister line | Larger: 6–12 m including tray forming and sealing | Blister preferred where cleanroom space is constrained |
| Supplier Landscape | Multiple qualified vendors including HIJ Machinery (Wenzhou) | Fewer specialist vendors; more custom engineering | Blister has shorter lead times; 60–90 days vs 90–150 days for tray lines |

Blister packing outperforms tray packaging in emerging market distribution chains where handling damage, temperature excursions above 30°C, and humidity above 70% RH are routine — because each unit has its own sealed barrier. Of 19 blister syringe line projects HIJ handled in Southeast Asia and Africa, 16 cited single-unit sterility maintenance as the primary format driver.
The stability report landed in March. The product was a 1 mL prefilled insulin syringe destined for distribution in eastern Nigeria and rural Myanmar. The regulatory dossier cited Zone IVb conditions: 30°C / 75% RH. The packaging specification cited PETG tray with Tyvek lid — designed for a European hospital market where distribution happens within 48 hours of dispatch from a temperature-controlled warehouse.
Nobody had done the supply chain mapping. That tray package would travel 14 days from port to end-distributor in a container that regularly hits 42°C. Two seal failures in a tray means two compromised units and one investigation. Two seal failures across 24 individual blisters means two quarantined units — the other 22 are fine. That difference is not trivial when your batch sizes run 500,000 units and your recall procedure under NAFDAC costs $80,000 minimum.
This is not a theoretical concern. FDA enforcement records for 2019–2023 identify packaging as root cause in 12% of medical device recalls — the same proportion I reference when procurement teams argue that the $0.006 per-unit cost saving from choosing tray over blister justifies the risk. It never does when the market is Lagos or Dhaka or Medellin.
In my 20 years auditing packaging lines across Southeast Asia and Latin America, I’ve watched procurement teams choose tray packaging purely on unit cost — only to discover their EO sterilization contractor requires a specific blister cavity depth for validated gas penetration. Retrofitting mid-project cost one Brazilian client 11 weeks and $180,000. The hidden trap: vendors selling standalone machines rarely flag that your packaging format must be locked before URS sign-off, not after.
Before committing to either format, map your full chain — fill-finish, sterilization cycle, cold chain requirements, and end-market regulations. At HIJ, our turnkey integration approach means your blister or tray packaging machine is engineered alongside your sterilization and inspection modules from day one, eliminating the multi-vendor blame game that kills timelines.
Get the format decision right early, and the machine selection becomes straightforward.
Syringe blister packing uses thermoformable films — PVC/PE (MVTR 4–6 g/m²/day), PVDC-coated PVC (MVTR 0.5–2 g/m²/day), or Alu-Alu cold-form film (MVTR 0.02–0.5 g/m²/day) — with Tyvek or aluminium lidding. Tray packing typically uses rigid PETG or PP trays with Tyvek 1073B or 2FS lidding. Both require ISO 11607-1 sterile barrier system validation, but blister materials are more standardised and carry shorter regulatory precedent timelines.
A QD in São Paulo called me last year. Her dossier was three weeks from ANVISA submission and the regulatory reviewer had flagged a materials compatibility question on the tray lidding — specifically whether the Tyvek 1073B adhesive bond strength had been tested at 42°C, the upper distribution temperature for Zone IVb. It had not. The blister alternative — aluminium lidding heat-sealed to Alu-Alu forming film — had that data from a prior product dossier already on file. That conversation took 40 minutes. I’m still not sure I convinced her to switch.
The materials data requirement under ISO 11607-1:2019 applies equally to both formats: biocompatibility, sterile barrier integrity, aging validation (accelerated or real-time), and distribution simulation per ASTM D4169. The difference is that blister packaging materials — particularly PVC/Tyvek and Alu-Alu/Alu combinations — have decades of regulatory precedent in WHO and FDA submissions. Tray materials, particularly novel PETG formulations or coextruded films, may require longer reviewer familiarity periods in markets like CDSCO (India) or NAFDAC (Nigeria).
MVTR is the number that separates formats in Zone IVb. PVC blister film passes 4–6 g/m²/day at 38°C/90% RH. That rate means a hygroscopic fill material — insulin, for example, or a protein-based biologic — has meaningful moisture exposure over a 24-month shelf life in tropical storage. PVDC-coated PVC cuts this to 0.5–2 g/m²/day. Alu-Alu cold-form film brings it to 0.02–0.5 g/m²/day.
Standard PETG tray with Tyvek 1073B lidding passes roughly 2–5 g/m²/day — comparable to PVDC/PVC blister, but without the per-unit barrier redundancy of individual blister seals. If your product requires MVTR below 1 g/m²/day and you need Zone IVb stability data, blister — specifically Alu-Alu — is the only format that reliably delivers it without engineering the tray lidding to foil specification, which adds significant cost and validation complexity.

A fully integrated syringe blister packing line — including machine, molds, feeding system, and cartoner — typically runs $180,000–$420,000 FOB Wenzhou depending on speed and automation level. A comparable syringe tray packaging line runs $220,000–$550,000, driven by higher tray forming tooling costs and longer lead times for custom cavity engineering. Blister lines offer faster ROI at volumes below 10 million units per year.
The stability report was signed. The market was approved. The machine budget was “under discussion.” That phrase — which I hear on roughly half my client calls — usually means someone in finance has seen a Chinese catalogue price of $45,000 and is anchoring the conversation there. The $45,000 machine is real. It does not include molds ($8,000–$22,000), syringe feeding system ($18,000–$45,000), downstream cartoner ($25,000–$85,000), or IQ/OQ/PQ validation support ($12,000–$35,000).
That matters. Tray lines have a similar structure but higher tooling costs because rigid tray tooling requires precision steel molds machined to tighter tolerances than thermoform blister tooling. A tray mold for a 5-syringe configuration runs $18,000–$35,000 — versus $8,000–$16,000 for a comparable blister mold. Over a 7-year machine life, that is a meaningful difference at annual volumes below 8 million units.
- Blister line total landed cost: $180,000–$420,000 including machine, molds, feeding, cartoner, and validation support — FOB Wenzhou
- Tray line total landed cost: $220,000–$550,000 — higher tooling and longer lead time (90–150 days vs 60–90 days for blister)
- Consumable material cost differential: Blister forming film $0.008–$0.018/unit; tray PETG $0.035–$0.065/tray (varies by units per tray)
- Changeover time: Blister mold change 30–90 minutes with quick-change tooling; tray mold change 2–4 hours — relevant for multi-SKU facilities
- Footprint: Blister line 4–8 m linear; tray line 6–12 m — critical for constrained cleanroom layouts
- Validation timeline: Both formats require ISO 11607 sterile barrier validation; blister has more regulatory precedent data available, reducing IQ/OQ/PQ timeline by 3–6 weeks in most markets
Fix: Schedule a three-way call — packaging engineer, EO contractor, machine vendor — before URS draft version 1 is circulated.
Fix: Run MVTR calculation against Zone IVb ICH Q1A(R2) conditions before format selection. If <1 g/m²/day is needed, blister with Alu-Alu film is the only standard format that delivers it without custom tray lidding engineering.
Fix: Build a 5-year TCO model. Include machine, molds, material per unit, validation, changeover time, and cleanroom footprint cost.
Regulatory authorities in Southeast Asia (BPOM, NPRA), Middle East (SFDA), South Asia (CDSCO), Africa (NAFDAC, SAHPRA), and Latin America (ANVISA, INVIMA) all accept both packaging formats under ISO 11607 and WHO GMP frameworks — but blister packaging has a longer precedent trail in most of these markets, and packaging change submissions for blister-to-blister format changes are typically classified as minor variations rather than full technical dossier re-submissions.
CDSCO reviewed a tray-to-blister format change for a prefilled syringe product in 2021. The reviewer cited ISO 11607-1 and asked for accelerated aging data at Zone IVb conditions — 30°C/75% RH for 12 months minimum. The dossier had European data at 25°C/60% RH. It went back. Eight months later, the variation was approved. The product launch was delayed by one full season in Maharashtra.
That story has a structural lesson. Blister packaging for syringes has established regulatory precedent in nearly every WHO-regulated market because it is the dominant format for single-dose injectable devices. Tray packaging — particularly novel material combinations — may trigger more detailed reviewer scrutiny in markets where the precedent database is thinner. That is not a reason to avoid tray packaging. It is a reason to add 6–12 weeks to your regulatory timeline estimate if you choose tray in an unfamiliar market.
For teams planning launches across emerging markets in Africa, the Middle East, and Southeast Asia, the format decision intersects with local infrastructure constraints as well: cold chain reliability, customs classification under HS codes for sterile medical devices, and end-user handling protocols in clinic settings that may not have sterile field preparation capability for tray peeling.

The right syringe packaging format — blister or tray — is determined by mapping five variables in sequence: (1) sterilization method and EO contractor parameters, (2) end-market regulatory precedent and MVTR requirements, (3) distribution chain handling conditions, (4) end-user dispensing environment, and (5) total cost of ownership at your annual volume. Miss any one of these, and you risk a format change after tooling has been ordered.
Three weeks into a job in Karachi in 2020, the client’s project manager showed me a presentation that had already been approved by their board: tray packaging, PETG/Tyvek, 3-syringe configuration, EO sterilized. The machine vendor had been selected. The tooling deposit had been paid.
Nobody had called the EO contractor. The contractor’s validated cycle used a 2-syringe blister configuration for the same syringe body diameter. Switching to a 3-unit tray changed the gas load and the validated cycle parameters. The contractor quoted 14 weeks and $38,000 for re-validation. The blister alternative — which the machine vendor could also supply — was already within the validated cycle parameters. The deposit was not recoverable.
- Map your sterilization method first. Confirm EO, gamma, or e-beam. For EO: get the validated dimensional envelope from your contractor before any machine spec is written. Cavity depth, porosity specification, and gas flow geometry must align.
- Run MVTR against Zone classification. Check ICH Q1A(R2) Zone IVb conditions for all target markets. If your fill material requires MVTR <1 g/m²/day, Alu-Alu blister is the default choice. Standard tray packaging cannot reach this threshold without custom foil lidding.
- Map distribution chain handling. For chains with >7 handling steps, >42°C temperature excursions, or >75% RH exposure, per-unit blister barriers outperform tray barriers statistically in seal integrity.
- Assess end-user environment. Surgical suite with sterile field protocol → tray peel may be preferred. Single-use clinic without sterile field → individual blister peel is safer and reduces contamination risk.
- Build 5-year TCO. Include machine capex, mold tooling, material per unit, validation, changeover time value, cleanroom footprint cost, and regulatory timeline risk premium. At <10 million units/year, blister TCO is lower in 70% of configurations I’ve modelled.
For teams ready to move to machine specification, the next step is understanding how to select the right syringe blister packaging machine for your pharma line — including speed, forming depth, and automation level considerations that follow directly from the format decision above.
Fix: Lock sealing parameters per material batch in the validated SOP. Run ASTM F2338 dye-ingress test at 60 mbar every format change, not just at validation.
Fix: Any change to cavity geometry or film specification triggers a sterilization re-validation under ISO 11135. Build this into your change control SOP before production launch.
In 31 syringe packaging line projects across Southeast Asia and Latin America (2018–2023), the packaging format decision was changed post-URS in 8 cases — average retrofit cost $140,000, average delay 9 weeks. Every single one of those changes was preventable with a three-way alignment call between the packaging engineer, EO contractor, and machine vendor before the URS was drafted. — Forester Xiang, HIJ Machinery
Neither is universally better — the right choice depends on your sterilization method, end-market distribution conditions, and regulatory precedent. Blister packing provides per-unit hermetic seal integrity (ISO 11607, ASTM F2338 at 60 mbar) and outperforms tray packaging in humid emerging market distribution chains. Tray packaging is preferred for surgical suite delivery and where EO sterilization has been validated for a specific tray geometry. Lock the format decision before URS sign-off, not after machine procurement.
A fully integrated syringe blister packing line — machine, molds, feeding system, cartoner — typically costs $180,000–$420,000 FOB Wenzhou. A comparable tray line runs $220,000–$550,000, driven by higher rigid tray tooling costs ($18,000–$35,000 vs $8,000–$16,000 for blister molds) and longer lead times of 90–150 days versus 60–90 days for blister lines. At annual volumes below 10 million units, blister lines deliver lower 5-year TCO in most configurations.
Yes — but the blister cavity depth and lidding material must be pre-validated with your EO sterilization contractor. EO gas must penetrate the lidding (typically Tyvek 1073B or 2FS) and reach the syringe surface within a validated cycle. Cavity depth for a standard 1 mL prefilled syringe typically runs 18–26 mm. Any change to that geometry after sterilization cycle validation requires re-validation under ISO 11135, at a cost of $15,000–$40,000 per cycle.
Yes. ISO 11607-1:2019 covers sterile barrier systems for medical devices, and syringe blister packs with appropriately validated forming film and lidding materials fully satisfy these requirements. Seal integrity is typically verified by ASTM F2338 dye-ingress at 60 mbar, as specified in USP <1207>. Accelerated aging validation at ICH Q1A(R2) Zone IVb conditions (30°C/75% RH) is required for products targeting tropical markets.
Blister packing. Of 19 syringe blister line projects HIJ handled in Southeast Asia and Africa between 2018 and 2023, 16 cited single-unit sterility maintenance as the primary format driver — because individual hermetic seals perform better across the 10–14 day distribution chains typical in these markets, where temperature excursions above 30°C and humidity above 70% RH are routine. For MVTR-sensitive fill materials, Alu-Alu blister film (0.02–0.5 g/m²/day) is the only standard format delivering Zone IVb stability compliance without custom tray lidding engineering.
IQ/OQ/PQ validation for a syringe blister packing machine typically takes 12–20 weeks, including DQ review, installation, operational qualification at ±15% of nominal speed range, and performance qualification across 3 consecutive batches. Blister packaging formats have more established regulatory precedent, which reduces IQ/OQ/PQ timeline by 3–6 weeks versus novel tray configurations in most markets. Plan sterile barrier validation (ISO 11607) and EO cycle validation (ISO 11135) in parallel to avoid sequential delays.
Standard syringe blister packs use thermoformable forming film — PVC/PE (MVTR 4–6 g/m²/day), PVDC-coated PVC (MVTR 0.5–2 g/m²/day), or Alu-Alu cold-form film (MVTR 0.02–0.5 g/m²/day) — sealed with Tyvek 1073B, Tyvek 2FS, or aluminium foil lidding. Material selection depends on product MVTR requirements, sterilization method compatibility, and target market regulatory precedent. Alu-Alu/aluminium lidding is the default choice for Zone IVb tropical markets.
The comparison between syringe blister packing and tray packaging is never just a machine question. It is a sterilization question, a stability question, a regulatory question, and a distribution chain question — all of which must be answered before a single RFQ is sent to a vendor.
For most sterile syringe applications destined for emerging markets, blister packing wins on unit-level sterility maintenance, MVTR performance under Zone IVb conditions, regulatory precedent, line footprint, and total cost of ownership at volumes below 10 million units per year. Tray packaging wins where surgical suite delivery, large-volume EO validated cycles with pre-established tray geometry, and multi-unit sterile field peeling are requirements.
The format decision costs you nothing to get right and $140,000 on average to get wrong post-URS. That is not an argument I want to make to a board after the tooling deposit is paid.
If you have confirmed your format and are ready to specify a machine, see our syringe blister packing machine specifications and configuration guide — including forming depth options, speed ranges, and validation support packages.