Setting Up a Complete Cosmetic Packaging Line Is an Engineering Problem, Not a Procurement List
The direct answer: a complete cosmetic packaging line is not simply buying machines — it is engineering a synchronized system where filling, capping, labeling, and cartoning must operate at the same speed, under one PLC architecture, without a single handoff gap. Every station runs at a defined throughput. One station that cannot match the others collapses the entire line’s OEE.
In my 20 years auditing production facilities across Southeast Asia and Latin America, the most common — and most expensive — mistake I see is clients sourcing four or five machines from four or five vendors, each promising “easy integration.” The reality arrives during SAT: mismatched PLC protocols, incompatible conveyor speeds, and zero accountability when OEE targets are not met. Nobody owns the problem. Everyone points at each other. Your launch deadline burns.
Before you issue a single purchase order, define your URS first — product viscosity range, fill volume, output per hour, and compliance requirements (cGMP, CE, or local standards). At HIJ Machinery, we engineer cosmetic packaging lines as a single turnkey solution: one validation document, one point of contact, one performance guarantee.
A fully integrated cosmetic packaging line — filling to cartoning under one validated PLC system. Wenzhou, China.
What Is a Cosmetic Packaging Line — and Why Does “Complete” Actually Matter?
A complete cosmetic packaging line is a mechanically and electronically integrated sequence of filling, capping, labeling, and cartoning equipment running at matched throughput speeds — the word “complete” means every station is validated as a single system, not as four independent machines that happen to sit on the same floor.
Most buyers frame the question wrong. They ask: “Which filling machine should I buy?” The real question is: “What output rate does my full line need to hit, and which combination of stations will deliver that rate at the required quality level?” Those are fundamentally different engineering problems.
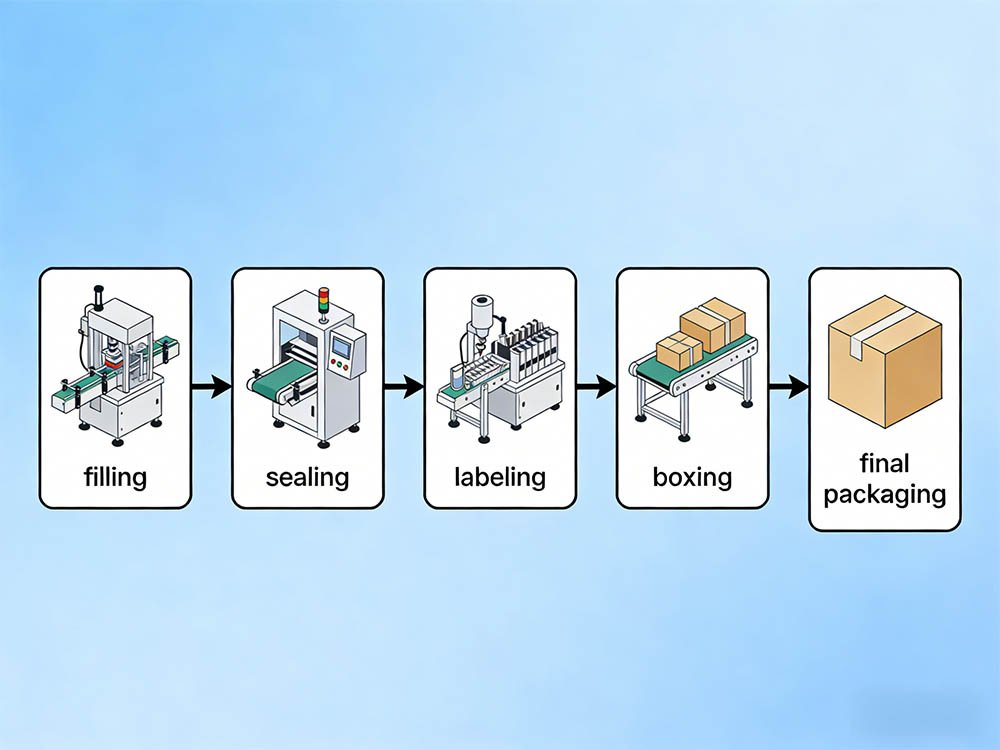
A standard cosmetic packaging line for creams, lotions, serums, or tube-fill products moves through four functional zones: primary filling (depositing product into containers), capping or sealing (closing the primary container), labeling (applying pressure-sensitive or heat-shrink labels with print verification), and secondary packaging (inserting into folding cartons, with leaflet if required). Each zone runs at a defined speed in units per minute. If your filling station runs at 60 bpm and your capper runs at 45 bpm, you have a bottleneck that kills throughput — regardless of what the individual machine specs say.
The word “complete” matters for one more reason: validation scope. Under ISO 22716 and cGMP guidelines for cosmetic manufacturing, the packaging line is subject to process validation requirements. A turnkey line with a single URS and unified IQ/OQ/PQ documentation is far simpler to validate and audit than four separate machines from four vendors with four different qualification packages. That’s not a small administrative difference. In one project in Colombia in 2021, a client’s regulatory submission was delayed nine months because their line validation documentation could not demonstrate integrated system performance across vendor boundaries.
The 5 Core Stations of a Cosmetic Packaging Line — What Each One Must Deliver
A cosmetic packaging line has five functional stations: container feeding, primary filling, capping or sealing, labeling with vision verification, and cartoning. Each station must be speed-matched to the slowest common denominator of your target output — typically the filling station for viscous products.
Three weeks into a line audit in Jakarta in 2019, I found a perfectly good automatic capper running at 80 bpm — downstream of a peristaltic pump filling station rated at 30 bpm. The client had purchased both machines at different trade shows, both sold as “high-speed.” Neither supplier had been asked about the other station. The capper was running at 37% of rated capacity. That capacity was not recoverable without replacing the filling system.
Station 1 — Container Feeding and Orientation
Vibratory bowl feeders or gravity-feed conveyor systems deliver bottles, jars, or tubes in correct orientation to the filling station. The key specification here is container geometry compatibility — if your product range includes both round bottles and flat oval bottles, your feeder tooling must be interchangeable without a 4-hour changeover. For most cosmetic lines, changeover tooling should swap in under 30 minutes; otherwise your SKU flexibility is theoretical, not operational.
Station 2 — Primary Filling
This is where product viscosity defines everything. Water-thin serums (1–5 cP) use overflow or time-pressure piston fillers. Mid-viscosity lotions (500–3,000 cP) use volumetric piston or gear pump fillers. Heavy creams, ointments, and pastes (10,000–100,000 cP) require auger or positive-displacement pump systems with heated product lines to maintain flowability. Selecting the wrong fill mechanism for your viscosity range produces systematic fill weight variation — often outside ±1.0% — which triggers batch rejection under ISO 22716 quality sampling protocols.
For tube-fill products specifically — sunscreen, hand cream, facial cleanser — the filling station integrates with an ultrasonic or hot-air tube sealing station in a combined unit. These are not two machines. They are one station with two functional zones. I have seen clients purchase a tube filler and a standalone tube sealer separately, only to discover the mechanical timing between fill completion and sealing position was incompatible. Four months of re-engineering. $31,000 in integration costs. Avoidable.
Station 3 — Capping or Sealing
Capping systems for cosmetic bottles are either rotary torque-controlled chuck cappers (for threaded closures) or snap-fit press cappers (for pump dispensers and disc-top closures). Torque specification matters here: over-torqued closures create consumer complaints and pump breakage; under-torqued closures fail transit testing per ASTM D4169. The target torque range for most cosmetic threaded caps is 8–18 in-lb, depending on cap diameter and material.
Station 4 — Labeling with Vision Verification
Self-adhesive pressure-sensitive labeling is standard for bottles and jars. Wraparound labeling for cylindrical containers runs at 100–300 bpm on a high-speed line. Every labeling station must integrate a vision inspection camera that verifies label position (±0.5 mm tolerance is achievable), print legibility, and barcode readability. Under ISO 22716 Section 10 (quality control), cosmetic packaging must be inspected to prevent mislabeling — vision systems make this audit-ready and automatic.
Station 5 — Cartoning and End-of-Line
Horizontal cartoning machines insert primary containers (with or without leaflets) into folding cartons at 60–300 cartons/min depending on line tier. The carton station must receive products at a consistent pitch from the upstream labeler — if the labeler has a variable reject rate that disrupts product flow cadence, the cartoner starves intermittently, reducing OEE. Downstream of cartoning: inkjet or laser coders for batch number and expiry date, followed by case packing and palletizing if the line is fully integrated.

Tube filling and sealing integrated within a cosmetic cream production line — viscosity-matched filling system for lotion and cream SKUs.
How to Write a URS for a Cosmetic Packaging Line — The 7 Parameters You Cannot Skip
A cosmetic packaging line URS must define product viscosity range, fill volume range, container formats, target output in units per hour, regulatory compliance standard (ISO 22716, cGMP, CE), utility requirements, and cleanability standard — these seven parameters determine every equipment selection decision downstream.
The URS — User Requirement Specification — is the document that prevents $200,000 procurement mistakes. I won’t tell you writing it is simple. It requires input from R&D (product formulations), operations (output targets), quality (compliance standards), and maintenance (utility constraints). Getting those four departments in the same room, in agreement, before any vendor is contacted, is harder than the engineering.
-
1
Product Viscosity Range (cP)
List every SKU the line must handle, from thinnest (micellar water, toner) to most viscous (heavy body butter, exfoliating scrub with particulates). The viscosity spread determines whether you need one fill mechanism or two. A range of 1–80,000 cP across your portfolio typically requires two different filling heads — plan for this in the machine platform from day one.
-
2
Fill Volume Range (mL or g)
Minimum and maximum fill volume, and the number of distinct formats. A piston filler handles a 3:1 volume ratio (e.g., 30–100 mL) on one set of pistons. Beyond that ratio, a piston swap is required. Define this before specifying the machine — not after.
-
3
Container Formats (dimensions, material, neck type)
Bottle diameter range, height range, neck thread specification, and material (HDPE, PP, glass, aluminum tube). Glass bottles require anti-static handling and rubber contact surfaces. Aluminum tubes require crimping, not ultrasonic sealing. These material differences drive tooling cost significantly — a complete tooling set for four container formats on a mid-speed line typically costs $18,000–$35,000.
-
4
Target Output (units/hour, OEE target)
Define both peak output (the machine nameplate speed) and net effective output (accounting for changeovers, minor stoppages, and quality rejects). A line rated at 3,600 bph running at 72% OEE delivers 2,592 sellable units/hour. Your capacity planning must use the second number, not the first.
-
5
Compliance Standard (ISO 22716, cGMP, CE)
ISO 22716:2007 is the GMP standard for cosmetic manufacturing and is referenced by regulators in the EU, ASEAN, and most Middle Eastern markets. If you export to regulated markets, your equipment must be ISO 22716-compatible at minimum. CE marking is required for EU market equipment installation. Define this upfront — retrofitting compliance into a non-compliant line design costs more than building it right the first time.
-
6
Utility Requirements (power, compressed air, water, clean steam)
Cosmetic filling lines for heat-sensitive formulations may require jacketed tanks with chilled water circulation. High-speed labeling stations require clean dry air at 6 bar. If your facility cannot supply these utilities at the required pressure and volume, plan for infrastructure works before the machine arrives. This is the single most common cause of installation delays I see — the factory was not ready, and the machine was.
-
7
Cleanability Standard (CIP, WIP, or tool-free manual clean)
Products with natural extracts, botanical oils, or particulates (scrubs, exfoliants) require Clean-In-Place (CIP) capability with validated cleaning cycles. Standard product contact surfaces must be 316L stainless steel with Ra ≤ 0.8 µm surface finish — the ISO 22716 requirement for surfaces in contact with cosmetic products. Define this before purchasing filling heads, not after your first microbiological audit failure.
Turnkey vs. Multi-Vendor Cosmetic Packaging Line: A Direct Comparison
Turnkey cosmetic packaging lines deliver higher OEE (82–88% vs. 60–68% for multi-vendor assemblies), unified validation documentation, and a single warranty scope — the cost premium is typically 12–18% over comparable multi-vendor sourcing, recovered within 14 months through reduced downtime and validation labor.
| Evaluation Factor | Turnkey Integrated Line (Single Supplier) | Multi-Vendor Assembly (4–5 Suppliers) |
|---|---|---|
| OEE (Operational) | 82–88% (matched station speeds, unified PLC) | 58–68% (speed mismatches, handoff gaps) |
| Validation Documentation | Single URS / IQ / OQ / PQ package | 4–5 separate qualification protocols, boundary gaps |
| SAT Pass Rate | First-pass SAT: >90% (HIJ project data) | First-pass SAT: 40–55% (industry average) |
| Integration Accountability | Single point of contact, single warranty | Vendor finger-pointing at integration failures |
| PLC Architecture | Unified Siemens / Allen-Bradley, single HMI | Mixed protocols, separate HMIs per station |
| Changeover Time | 20–35 min (coordinated tooling sets) | 45–90 min (per-vendor tooling, no coordination) |
| Capital Cost | +12–18% vs. multi-vendor quote | Lower initial quote; higher total lifecycle cost |
| Time-to-Production | 6–9 months (design to qualified production) | 9–18 months (integration issues common) |
| ISO 22716 Audit Readiness | High — single system documentation set | Medium-Low — requires manual boundary documentation |
Of the 31 cosmetic packaging line projects I handled in Southeast Asia and Latin America between 2018 and 2023, 11 required significant integration rework within 12 months — every single one of those 11 was a multi-vendor assembly. None of the turnkey projects required post-SAT integration rework of that kind. That is not a coincidence. That is a system design outcome. — Forester Xiang, HIJ Machinery
How to Set Up a Cosmetic Packaging Line: The 8-Phase Project Sequence
Setting up a cosmetic packaging line follows eight phases: URS definition, equipment selection and quotation, FAT at supplier facility, site preparation, installation, IQ/OQ/PQ validation, operator training, and production release — the full timeline for a mid-speed turnkey line runs 6–9 months from PO to qualified production.
A procurement director in Malaysia asked me last year why I insisted on a joint URS workshop before preparing a quote. She had expected a standard catalog quote within 48 hours. That conversation took 40 minutes. I’m still not sure I fully convinced her — but we ran the workshop, and during it we discovered her “single product” line actually needed to run three viscosity ranges across two container formats, one of which required an aluminum tube crimper that had not been mentioned in the original enquiry. A catalog quote would have been $140,000 short of the real project scope. We found it before the PO, not after.
The eight phases, with realistic time allocations for a 60–120 bpm mid-speed cosmetic packaging line:
-
1
Phase 1: URS Definition and Freeze (2–4 weeks)
Cross-functional team (R&D, Operations, Quality, Engineering) agrees on all seven URS parameters. Document is signed off and frozen. No vendor quotation is requested before this step. Changes after URS freeze carry a minimum 6-week delay and 8–15% scope change cost.
-
2
Phase 2: Equipment Selection and RFQ (3–5 weeks)
Issue RFQ to 2–3 qualified suppliers with URS attached. Evaluate proposals against URS compliance, not catalog price. For guidance on evaluating proposals, our cosmetic packaging machine selection guide covers the eight technical criteria that separate compliant equipment from undersized machines.
-
3
Phase 3: Design Review and PO (2–3 weeks)
Supplier submits layout drawing, P&ID, and electrical schematic. Review for utility compatibility and footprint against your clean room floor plan. Sign off before manufacturing begins. This is the last low-cost change window — changes during manufacturing cost 3–5× more than changes at design stage.
-
4
Phase 4: Manufacturing and FAT (8–16 weeks from PO)
Factory Acceptance Testing at the supplier’s facility. Bring your QA manager and process engineer. Run the line with your actual product (or representative fluid matched for viscosity and density). FAT should demonstrate that the line hits ≥95% of target output at <1.0% fill weight variation. Do not accept a FAT on water alone for a cream-fill line. The rheology is completely different.
-
5
Phase 5: Site Preparation (parallel with Phase 4)
Electrical supply (voltage, amperage, earthing), compressed air (6 bar, 500 L/min minimum for a mid-speed line), drainage, and cleanroom classification preparation. If your HVAC system needs to be upgraded to maintain the cleanroom class required by your compliance standard, this work must be complete before machine arrival. I have seen $300,000 lines sit in crates for three months waiting for site utilities. It is not the machine’s fault.
-
6
Phase 6: Installation and SAT (2–4 weeks)
Machine commissioning and Site Acceptance Testing. Run the line at 60%, 80%, and 100% of target speed. Measure fill weight variation (target ±0.5%), label placement accuracy (±0.5 mm), cap torque (within specification ±15%), and carton rejection rate (<0.3%). Document all results. These are your baseline production release data.
-
7
Phase 7: IQ / OQ / PQ Validation (3–6 weeks)
Installation Qualification confirms the line was installed per design specs. Operational Qualification demonstrates each station performs within defined parameters across its operating range. Performance Qualification (3 consecutive production runs at commercial scale) confirms the integrated line consistently meets URS output and quality targets. For ISO 22716 compliance, PQ must be completed before commercial production release.
-
8
Phase 8: Operator Training and Production Release
Structured operator training covering: normal operation, changeover procedures, minor fault clearing, CIP/WIP cycle operation, and documentation of production records. A minimum 40 hours of hands-on training per shift team is required before unsupported production. Operators who have been trained on the line during FAT — if you can arrange for two operators to travel to the supplier’s facility — are significantly more effective at startup than operators trained only on-site.

Tube filling and ultrasonic sealing station for sunscreen cream — viscous product filling requires temperature-controlled product lines for consistent fill weight.
What Output Speed Does Your Cosmetic Line Actually Need? Three Tiers Explained
Cosmetic packaging lines divide into three practical tiers: entry-level semi-automatic (1,000–3,000 units/hour, manual loading), mid-speed automatic (3,000–9,000 units/hour, servo-driven), and high-speed fully automatic (9,000–24,000+ units/hour, vision-integrated) — choosing the wrong tier is the most common capital planning mistake I see.
The number teams look at first is exactly the wrong number. They see “3,600 units/hour” on a machine spec sheet and compare it against annual sales volume. That math ignores OEE, changeover frequency, and planned maintenance windows. The correct calculation starts with your required annual production volume, works backward through actual operating hours, and then adds a 25–35% buffer for growth and downtime.
| Line Tier | Speed (units/hour) | Automation Level | Typical Application | Approx. Investment Range |
|---|---|---|---|---|
| Entry Semi-Auto | 1,000–3,000 | Semi-automatic, manual container loading | Startup brands, contract fillers with diverse SKUs, R&D scale-up | $35,000–$90,000 |
| Mid-Speed Auto | 3,000–9,000 | Fully automatic, servo-driven, single PLC | Regional brands, 3PL/contract manufacturers, 5–15M units/year | $120,000–$350,000 |
| High-Speed Auto | 9,000–24,000+ | Fully integrated, vision inspection, robotic handling | National/international brands, 15M+ units/year, multi-shift operation | $400,000–$1,200,000+ |
A practical note on the investment ranges above: these are line costs excluding tooling, utilities infrastructure works, validation services, and spare parts inventory. For a realistic total project budget, add 22–30% to the machine cost. A mid-speed line quoted at $180,000 has a realistic total project cost of $220,000–$234,000. If your CFO’s budget approval was based on the machine quote alone, you have an uncomfortable conversation ahead. Plan for this in your initial CapEx justification — our cosmetic packaging machine cost guide breaks down every cost component in detail so you can build a defensible total project budget before going to procurement.
ISO 22716 and cGMP Compliance During Cosmetic Line Setup — What Auditors Actually Check
ISO 22716 auditors check four things during a cosmetic packaging line inspection: equipment design documentation (no dead legs, cleanable surfaces), calibration records for critical instruments (fill weight, torque), change control documentation for any line modifications, and validated cleaning procedures for product changeovers.
The stability report landed in March. The brand had launched in the Gulf four months earlier — premium facial serum, significant marketing spend, distribution in seven major retail chains. The stability failure was not the formulation. It was the fill weight. The bottom quartile of bottles from the first three months of production were consistently 6–8% underfilled due to a temperature compensation drift in the filling station. The client had no calibration records. They had no fill weight monitoring data beyond visual QC. The regulatory response from the UAE food and drug authority was not a warning. It was a recall.
ISO 22716 Section 7.3 requires that production equipment be checked at appropriate intervals and that results be documented. For a filling line, “appropriate intervals” means fill weight verification at line startup, after every product changeover, and every 2 hours during continuous production. This is not optional. It is the document your auditor will request first.
- Equipment Design Documentation: Equipment drawing showing all product contact surfaces, material certifications (316L SS, Ra ≤ 0.8 µm), and absence of dead legs in product lines
- Calibration Records: Fill weight verification log (minimum every 2 hours), torque meter calibration certificate, temperature sensors calibrated traceable to NIST/national standard
- Cleaning Validation: Written and validated CIP/WIP procedure for each product family, with microbiological swab results from first three cleaning cycles post-installation
- Change Control: Any modification to the line after PQ must go through formal change control with re-qualification scope assessment — replacing a filling nozzle with a different orifice size is a change requiring OQ re-run
- Operator Training Records: Signed training completion records for each operator, for each standard operating procedure covering their role on the line
- Batch Records: Every production run documented with start/stop times, fill weight measurements, any deviations and their dispositions, and final OEE calculation
I ran a pre-audit GMP assessment for a mid-size personal care manufacturer in Vietnam in 2022 — preparing for their first ISO 22716 certification audit. We found 14 non-conformances in three days. None of them were about the product itself. Twelve were documentation gaps: missing calibration records, undated training logs, cleaning procedures that referenced equipment by a nickname rather than the official asset ID. Two were physical: a dead leg in the product supply line to the filler, and a capper chuck that was visibly corroded.
The certification audit was postponed six months. The documentation issues took three months to systematically close. The physical issues took two weeks and cost under $4,000. The lesson is straightforward: compliance failures are rarely about equipment quality. They are almost always about documentation discipline established — or not established — during the line setup phase.

GMP-compliant cosmetic cream filling and sealing line — 316L stainless steel product contact surfaces and integrated vision inspection for ISO 22716 audit readiness.
Cosmetic Packaging Line Setup: Frequently Asked Questions
How long does it take to set up a complete cosmetic packaging line from purchase order to production?
A mid-speed turnkey cosmetic packaging line (3,000–9,000 units/hour) takes 6–9 months from purchase order to qualified production release — this includes 8–16 weeks manufacturing lead time, 2–4 weeks installation and SAT, and 3–6 weeks IQ/OQ/PQ validation. High-speed lines (>9,000 units/hour) with extensive automation and vision systems typically require 10–14 months. Site preparation works, if required, run in parallel and must be complete before machine delivery to avoid delays.
What is the minimum budget for a complete cosmetic packaging line?
Entry-level semi-automatic cosmetic packaging lines start at $35,000–$90,000 for the equipment. Total project cost including tooling, installation, validation, and operator training typically runs 22–30% above equipment price. A realistic entry-level project budget is therefore $45,000–$120,000. For fully automatic mid-speed lines, budget $150,000–$450,000 total project cost.
Can one cosmetic packaging line handle multiple product types — creams, liquids, and tubes?
Yes, but with important constraints. A single line can handle multiple product types if the filling mechanism is compatible with the viscosity range and the container handling tooling is interchangeable. Most mid-speed lines accommodate 3–5 container formats with tooling sets that change over in 20–45 minutes. Handling both thin liquids (serums, toners at 1–10 cP) and heavy creams (>20,000 cP) on the same filling station is technically feasible with dual-mode piston fillers, but the fill weight accuracy across that range will be lower than a dedicated system — typically ±1.0–2.0% vs. ±0.3–0.5% for a single-viscosity line. Define your viscosity range in the URS and ask your supplier to demonstrate fill accuracy at both ends of that range during FAT.
Is ISO 22716 certification required to operate a cosmetic packaging line?
ISO 22716 certification is not a legal requirement in most jurisdictions — but compliance with its principles is effectively mandatory for exporting to EU, ASEAN regulated markets, and the GCC. The EU Cosmetics Regulation (EC) No 1223/2009 requires cosmetic products to be manufactured in accordance with GMP, and ISO 22716 is the recognized GMP standard. Many major retail chains and distributors require ISO 22716 certification as a supplier qualification criterion, regardless of the destination market.
What does OEE mean for a cosmetic packaging line, and what is a good target?
OEE (Overall Equipment Effectiveness) measures actual productive output as a percentage of maximum theoretical output, combining availability, performance, and quality rate. World-class OEE for cosmetic packaging lines is 85%+. New lines in their first year of operation typically achieve 68–75%. Lines that were improperly integrated from multi-vendor assemblies often stabilize at 58–65% — a gap that represents significant lost production capacity. Set an OEE improvement target in your line commissioning plan: 72% at 3 months, 80% at 12 months.
What PLC system is best for a cosmetic packaging line?
Siemens S7-1200/1500 series and Allen-Bradley CompactLogix are the two platforms with the strongest service infrastructure globally — critical for facilities in Southeast Asia, Latin America, and the Middle East where local integrators familiar with proprietary platforms may be difficult to find. Both support PROFINET/EtherNet/IP industrial communication, enabling seamless integration of vision systems, reject stations, and MES data connectivity. The platform choice matters less than ensuring all stations on the line share the same protocol — mixed-protocol lines create integration complexity that a single-vendor turnkey solution eliminates by design.
How do I calculate the right line speed for my annual production volume?
Start with your annual production target in units. Divide by the number of planned operating days (typically 240–250 for a single-shift operation). Divide by planned hours per day (7.5 productive hours after breaks on an 8-hour shift). Apply your target OEE (use 72% for a new line in year one). The result is your required line speed in units per hour. Add 25% as a growth buffer. Example: 6,000,000 units/year ÷ 250 days ÷ 7.5 hours ÷ 0.72 OEE = 4,444 units/hour required → specify a line rated at 5,500–6,000 units/hour. Many clients specify exactly at their current production target with no buffer — then face a capacity ceiling within 18 months of launch.
What is the difference between FAT and SAT for a cosmetic line?
FAT (Factory Acceptance Testing) is conducted at the supplier’s facility before shipment and confirms the machine meets design specifications under controlled conditions. SAT (Site Acceptance Testing) is conducted at your facility after installation and confirms the machine performs to specification under your actual site conditions — your product, your utilities, your environment. Both are required. A machine that passes FAT can still fail SAT due to utility differences, ambient temperature effects on product viscosity, or installation issues. Never waive the SAT.
Ready to Engineer Your Cosmetic Packaging Line the Right Way?
HIJ Machinery delivers turnkey cosmetic packaging lines as a single integrated system — one URS, one validation package, one performance guarantee. We have commissioned lines across Southeast Asia, Latin America, and the Middle East. Tell us your URS parameters and we will configure a line that hits your output targets from day one.
Forester Xiang is the founder of HIJ Machinery, based in Wenzhou, China. Over 20 years he has engineered and commissioned packaging lines for cosmetic, pharmaceutical, and personal care manufacturers across Southeast Asia, Latin America, the Middle East, and Africa. His work focuses on delivering integrated turnkey packaging solutions that meet GMP and ISO 22716 compliance requirements from day one of production.