🎯 Buyer's Intelligence Report
How to Evaluate Blister Packaging Machine Suppliers — Before the Contract Locks You In
Most buyers discover supplier problems after installation. By then, a missing IQ protocol costs 6–8 months. A proprietary PLC costs $40,000 in sole-source service fees. The shortcut of skipping a witnessed FAT costs a batch recall. This guide tells you exactly what to demand — and exactly what to walk away from — before you sign anything.
✅ Direct Answer
A qualified blister packaging machine supplier must demonstrate five non-negotiable criteria: complete cGMP documentation package (IQ/OQ/PQ templates included), witnessed FAT at the manufacturing facility, open PLC architecture with no proprietary software lock-in, a named after-sales engineer with direct contact, and turnkey integration accountability across blister, cartoner, and checkweigher interfaces. Suppliers who cannot satisfy all five carry quantifiable project risk — measured in months of lost production and six-figure remediation costs.
6–8 mo
Average delay when IQ/OQ documentation is missing post-installation
12%
Of pharmaceutical recalls traced to packaging root cause (FDA 2019–2023)
$40k+
Typical sole-source service fee from proprietary PLC lock-in, year 2–3
31
Blister line projects in Southeast Asia reviewed by HIJ (2018–2023)
Why Most Blister Packaging Machine Supplier Evaluations Fail Before They Start
A procurement director in Kuala Lumpur called me in 2021 — three months after commissioning. Wrong call time to make. The machine was running. But the IQ documentation had gaps. The CDSCO dossier reviewer had flagged it. The product launch was postponed by seven months.
That conversation lasted 40 minutes. I'm still not sure I convinced him that the documentation problem was fixable faster than starting the dossier over. What I am sure of: none of it would have happened if the RFQ had included a one-page documentation requirement before the supplier was shortlisted.
Most blister packaging machine supplier evaluations fail for one reason — they start at price. The budget gets set, three quotes come in, the lowest gets shortlisted. Nobody asked about IQ/OQ templates. Nobody asked about PLC architecture. Nobody asked who the after-sales engineer is. Those questions arrive later. By then, the contract is signed.
⚠️ Field Warning
FDA enforcement records for 2019–2023 show packaging as the root cause in 12% of pharmaceutical recalls — the category with the strongest correlation to tropical market distribution failures. When I use that number in conversations with procurement teams who tell me the cost saving of $0.006 per blister card justifies the documentation risk, the math changes fast.

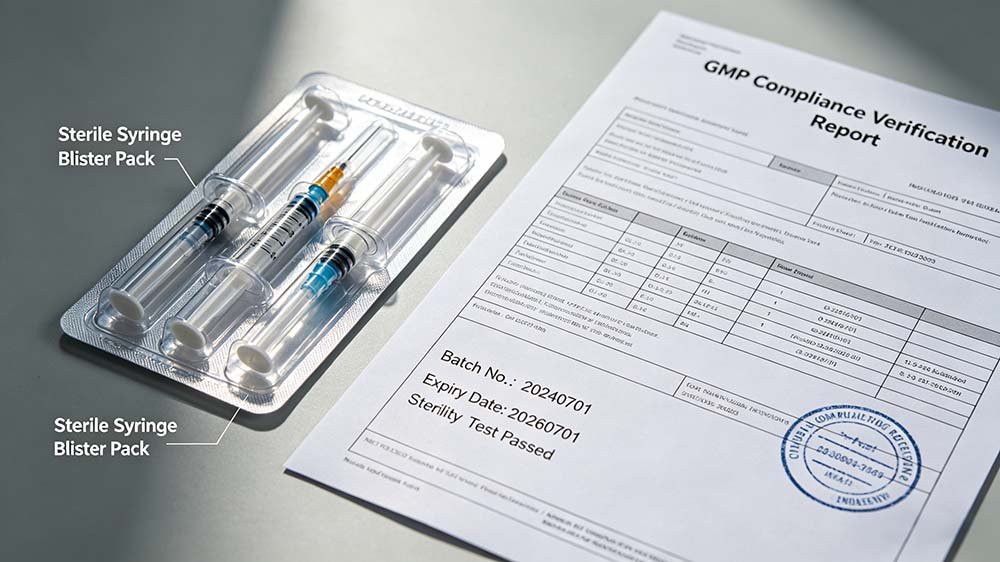
Pre-purchase technical documentation review — the step most buyers skip and later regret.
The 5-Criteria Framework for Evaluating Blister Packaging Machine Suppliers
A qualified blister packaging machine supplier meets five criteria simultaneously — not four, not three. Each criterion maps to a specific failure mode I've seen in the field. Skip one, and you're accepting a known risk without a price for it.
| Criterion | What to Demand | Red Flag | Failure Mode if Skipped | Risk Level |
|---|---|---|---|---|
| 1. cGMP Documentation | IQ/OQ/PQ template set, FAT checklist, DQ report, material contact certifications (316L stainless, FDA-grade seals) | “We can provide documentation after installation” | Dossier rejection, 6–8 month delay | ❌ Critical |
| 2. Witnessed FAT | Live run at supplier facility, your product or surrogate, your film specification, your speed target | FAT offered by video call only | Hidden forming defects, incorrect tooling discovered post-SAT | ❌ Critical |
| 3. Open PLC Architecture | Standard Siemens/Allen-Bradley/Omron PLC, full ladder logic access, no encrypted proprietary modules | “Software is proprietary for IP protection” | $40,000+ sole-source service lock-in years 2–5 | ⚠️ High |
| 4. Named After-Sales Engineer | Direct mobile number, maximum 24-hour response SLA in writing, remote access capability | “Contact our service department” (generic) | 72+ hour downtime during critical batch runs | ⚠️ High |
| 5. Turnkey Integration Accountability | Single supplier accountable for blister-to-cartoner interface, communication protocol documentation, line speed matching | “Cartoner integration is your responsibility” | Multi-vendor blame loop, 3–6 month integration delay | ⚠️ High |
🧠 Forester's Insight — 20 Years in the Field
The Direct Answer: Why Supplier Selection Is a Post-Installation Problem
Choosing the right blister packaging machine supplier is not about price. It is about who stands behind the machine when your production line stops at 2 a.m. during a critical batch run.
In 20 years auditing facilities across Southeast Asia and Latin America, I've watched buyers get burned by the same mistake: selecting a supplier based on a polished brochure, only to discover post-installation that the machine lacked proper cGMP documentation, couldn't pass SAT validation, or used proprietary PLC protocols that locked them into expensive sole-source service contracts. The hidden trap? A “competitive” quote rarely includes the true cost of multi-vendor integration chaos. When your blister machine, cartoner, and checkweigher refuse to communicate, you pay for the systems integrator.
Before signing anything: demand a structured URS review, witness a live FAT at the supplier's facility, and verify their after-sales engineering team is reachable — not just their sales rep. At HIJ, our turnkey integration philosophy means one accountable partner from machine commissioning through compliance sign-off, eliminating the blame-game that costs buyers months of lost production.
Write the URS Before You Send a Single RFQ
Of the 31 blister line projects I handled in Southeast Asia between 2018 and 2023, 11 required a forming material change after month-12 stability failure. Nine of those eleven had not specified forming film MVTR requirements in the original URS. The supplier delivered exactly what was asked for. The problem was what wasn't asked.
A User Requirement Specification is not a wish list. It's a legal and technical boundary document. A supplier who cannot respond to a structured URS line by line — with specific technical answers, not marketing language — is not ready to supply pharmaceutical equipment. Full stop.
What a Pharmaceutical-Grade URS Must Cover
- Product specification: tablet/capsule dimensions (length × width × height in mm), weight range, shape, surface fragility index, maximum acceptable breakage rate (<0.1% recommended)
- Forming material: PVC thickness (μm), PVDC coating weight (g/m²), or cold-form Alu-Alu specification including MVTR target (<0.5 g/m²/day for hygroscopic APIs)
- Output target: blisters per minute at stated efficiency ≥85% OEE, including format changeover time (<30 minutes for multi-SKU lines)
- Regulatory market: WHO GMP, 21 CFR Part 211, EU GMP Annex 1 — each imposes different documentation, material, and validation requirements
- Utility requirements: compressed air pressure (typically 6–8 bar), power supply (3-phase, 380V/50Hz or 460V/60Hz), chilled water for cold forming
- Integration interfaces: upstream filling/counting equipment protocol, downstream cartoner communication, checkweigher rejection signal type
- GMP construction: contact parts material (316L stainless minimum), surface roughness (Ra ≤0.8 μm for pharmaceutical contact surfaces), cleanability
🛠️ Diagnostic
Supplier responds to your URS with a brochure instead of a technical compliance matrix
This is not a communication style difference. It is a capability signal. A supplier who cannot complete a URS compliance matrix line by line — marking each item as “compliant,” “compliant with deviation,” or “not compliant + proposed alternative” — has not supplied pharmaceutical equipment to a dossier-reviewed facility before. Walk away, or require a documented demonstration run before advancing. At minimum, do not pay a deposit until the matrix is complete and signed.

A completed URS compliance matrix separates qualified blister packaging machine suppliers from catalog sellers.
The FAT Protocol: What a Witnessed Factory Acceptance Test Actually Proves
A QA director in São Paulo called me last year. They'd accepted FAT by video call. “The machine looked fine,” she said. Six weeks after installation, the forming station was producing pockets 0.4mm shallower than the cavity spec — well within normal tablet insertion tolerance for the demo product used on the video call, but outside spec for her 8mm capsule. The tooling needed rework. Eight weeks offline.
A video call FAT proves the machine powers on. That's it. A witnessed FAT at the supplier's facility proves something useful — that your specific product, in your specific film specification, at your target speed, produces blister cards that pass your acceptance criteria. Nothing else does.
FAT Acceptance Criteria Checklist
- Run duration: minimum 2-hour continuous production run at rated speed, no manual interventions, witnessed by your QA representative
- Sealing integrity: 100% of sampled blisters pass dye-ingress test per ASTM F2338 at 60 mbar differential — the method specified in USP <1207> for container closure integrity
- Forming depth consistency: cavity depth tolerance ±0.2mm across all cavities, measured at start, middle, and end of 2-hour run
- Product integrity: zero broken tablets/capsules in 1,000-blister sample, verified visually and by weight check
- Alarm and rejection system: intentionally introduce 3 empty cavities — all must be detected and rejected within specification
- Format changeover time: witnessed changeover from Product A format to Product B format — record actual time vs. URS requirement
- Documentation delivery: FAT report, signed IQ protocol template, spare parts list with lead times, and PLC access credentials handed over before machine ships
Accepting a video-call FAT is not a time-saving shortcut — it is a documentation liability that your regulatory reviewer will identify before your dossier reviewer does. Every project I've managed where FAT was witnessed in person produced fewer post-installation surprises. The correlation is not coincidental. — Forester Xiang, HIJ Machinery
PLC Architecture: The $40,000 Question Nobody Asks Until Year Three
The procurement director showed me the sheet. Machine A: $0.006 cheaper per blister card in consumable film cost. Annual volume: 8 million cards. Total annual saving: $48,000. I asked what a batch recall cost. She didn't have the number. I told her: in our region, the average pharmaceutical batch recall costs $1.2 million to $3.8 million when you include market withdrawal, regulatory response, and product replacement.
Then I asked a different question. What PLC does Machine A run?
Proprietary. Encrypted. Software updates through the manufacturer only. Service engineer on-site rate: $2,400 per day, minimum 5-day call-out, 21-day average lead time from Asia. Year-one service contract: $38,000. Year-two escalation clause: 15%.
That's the real cost. The film saving was gone in the first service call.
Acceptable vs. Unacceptable PLC Configurations
| PLC Type | Acceptable for Pharma? | Service Cost Profile | 21 CFR Part 11 Compatibility |
|---|---|---|---|
| Siemens S7-1200 / S7-1500 (open access) | ✅ Preferred | Local integrator support globally, $80–$150/hr | Full audit trail native |
| Allen-Bradley / Rockwell (open access) | ✅ Preferred | Widest global support network | Full audit trail native |
| Omron NX / NJ series (open access) | ✅ Acceptable | Strong Asia-Pacific support | Audit trail with configuration |
| Generic Chinese PLC (Wecon, Xinje) — open | ⚠️ Low-risk markets only | Low cost, limited global support | Manual audit trail, 21 CFR risk |
| Any proprietary encrypted PLC | ❌ Not recommended | Sole-source, $2,400+/day on-site | Audit trail not user-controllable |
📋 Compliance Reference
21 CFR Part 11 (Electronic Records, Electronic Signatures) requires that computerized systems used in pharmaceutical manufacturing maintain secure, computer-generated audit trails that independently record the date and time of operator entries and actions that create, modify, or delete electronic records. A proprietary PLC where the supplier controls software access does not give the facility operator independent control of that audit trail — a deficiency FDA inspectors have cited in warning letters for blister packaging line operations.
After-Sales Accountability: What “24/7 Support” Actually Means in Practice
I saw this in a plant outside Pune in 2019. 2:40 a.m. The sealing station had thrown a thermal fault. Production was stopped. Three batches of a time-sensitive API product were sitting in the filling room — temperature-controlled, on a clock. The QA manager called the supplier's service hotline. He got a voicemail. He called again. A different voicemail. He found my number from a trade show card in his desk.
That thermal fault was a PID controller setting issue. Fixable in 12 minutes with remote access. Without remote access, the machine sat until the next business day in Shanghai. The product didn't make it.
“24/7 support” means nothing unless you can verify: who specifically responds, how fast, with what access level, and under what contractual obligation.
After-Sales Support Verification Checklist
- Named engineer assignment: a specific person's name, direct mobile number, and email — not a department inbox
- Response SLA in writing: maximum 2 hours for remote diagnosis, 24 hours for on-site dispatch decision — this goes in the commercial contract, not the service brochure
- Remote access protocol: VPN or TeamViewer access to PLC and HMI, encrypted, auditable, compatible with your facility's IT security policy
- Critical spare parts on-site: supplier specifies the minimum local spare parts inventory required — heating elements, sealing rollers, PVC forming plugs, drive belts
- Field engineer timezone coverage: for markets in Southeast Asia, Africa, or Latin America, verify the supplier has a regional service office or an authorized service partner within 4 hours' flight
🛠️ Diagnostic
Supplier cannot name a specific after-sales engineer at time of quote
Ask the sales representative: “Who will be our named technical contact after commissioning? Can I have their direct phone number today?” If the answer is “we will assign someone after installation,” that answer tells you the after-sales structure does not yet exist. It will be assembled reactively after a problem occurs, not proactively before one.
I won't tell you this is a dealbreaker for every budget scenario — some small-volume buyers in developing markets genuinely cannot access tier-1 service. But in those cases, the risk must be priced explicitly: on-site spare parts budget, local electrician retainer, and a 10% production contingency in year one.

Named after-sales engineer on-site at client facility — the accountability standard buyers should demand before signing.
Turnkey Integration: Why Single-Supplier Accountability Changes the Risk Equation
The stability report landed in March. The product had been running for six months. Seal integrity on months 7–9 was below specification — not by much, 0.3 g/m²/day above the threshold. The blister machine supplier said it was a film issue. The film supplier said it was a sealing temperature variance. The cartoner manufacturer said nothing, because nobody had asked them yet. The client's engineering team spent four months running experiments that should have taken three weeks with a single accountable supplier.
Turnkey integration accountability does not mean buying every machine from the same catalog. It means one supplier who contractually owns the interface between machines — blister former, cartoner, checkweigher, case packer — and who cannot redirect fault to another vendor. That single sentence is worth more than any feature comparison table.
For buyers evaluating a pill blister packaging machine as part of a broader line expansion, the integration question is where the supplier's real capability becomes visible. Any manufacturer can sell a blister machine. Not many can commission a blister machine, a cartoner, and a checkweigher as an integrated system that passes line qualification in a single IQ/OQ/PQ cycle.

A single-supplier turnkey line eliminates the multi-vendor blame loop — the most expensive hidden cost in pharmaceutical packaging projects.
Questions to Test Integration Capability
- “Can you provide a line speed matching calculation showing the blister machine output (blisters/min) is compatible with your cartoner input (cartons/min) at our SKU mix?”
- “Who is contractually responsible if the checkweigher rejection signal causes a blister machine jam — you or the checkweigher manufacturer?”
- “Show us a previous installation where you completed a single IQ/OQ/PQ cycle covering blister machine and cartoner as an integrated system. Reference site, contact name, and validation date.”
- “What SCADA or line control software links your blister machine data to the plant MES or batch record system?”
Building a Supplier Shortlist: A Step-by-Step Process for Pharmaceutical Buyers
The number teams look at first is exactly the wrong number. Unit price lands on page one of every proposal. The variables that determine total cost of ownership over a 10-year equipment lifecycle — documentation quality, service response time, PLC openness, spare parts availability — are buried in the technical annex, if they exist at all. Here is the process I give to procurement teams who are building a supplier shortlist for the first time.
-
1
Issue a structured RFQ with URS attached
Do not issue a price-only RFQ. Attach your URS and require a line-by-line compliance matrix as part of the response. Any supplier who does not return a completed matrix within your RFQ deadline is self-disqualifying.
-
2
Score suppliers on the 5-criteria framework, not price
Use the five criteria from the table above. Assign a pass/fail to criteria 1 and 2 (documentation and FAT). Score criteria 3–5 on a 1–5 scale. Only shortlist suppliers who pass both binary criteria. This typically eliminates 40–60% of respondents immediately.
-
3
Request reference contacts, not reference letters
Ask for the direct phone number of a QA manager at a reference installation that completed IQ/OQ/PQ within the last three years. Call them. Ask two questions: “Did the documentation match what was promised?” and “How long did it take to reach a live engineer after your first post-commissioning fault?” Those answers tell you everything a brochure cannot.
-
4
Conduct a witnessed FAT with your actual product
Ship surrogate product or placebo tablets to the supplier's facility. Run a 2-hour witnessed FAT. Bring your QA engineer, not just your procurement manager. Evaluate against the FAT acceptance criteria checklist above — in writing, signed by both parties.
-
5
Negotiate documentation and service terms before price
Finalize what the IQ/OQ/PQ package contains, who the named service engineer is, and what the response SLA is — before you negotiate the final price. Suppliers who resist documentation terms before a price discussion are telling you their documentation is not ready.
-
6
Include a 12-month performance guarantee clause
Contract for: machine availability ≥85% OEE in months 3–12, seal integrity pass rate ≥99.8% on production samples, and response time SLA (2 hours remote, 72 hours on-site for markets within 4 hours flight of a regional office). Tie 10–15% of final payment to 90-day post-commissioning performance data.
🛠️ Diagnostic
Supplier asks you to sign a deposit before completing URS review or before FAT date is confirmed
This sequence matters. A deposit signals commitment to a machine specification. If URS review is incomplete when the deposit is paid, the supplier has no contractual obligation to change the specification. I've seen buyers pay a 30% deposit and then discover the forming tooling pitch doesn't match their capsule size. The supplier wasn't dishonest — the specification simply hadn't been agreed. Pay nothing until the URS compliance matrix is signed, the FAT date is confirmed, and the documentation package list is contractually defined.
Regional Blister Packaging Machine Supplier Landscape: What Buyers in Asia, Africa, and Latin America Need to Know
The supplier evaluation framework above is universal. The regional context is not. A buyer in Manila sourcing for a WHO-PQ product faces different documentation requirements than a buyer in Lagos sourcing for a NAFDAC-registered product, or a buyer in Bogotá preparing for INVIMA approval. The machine specification may be identical. The compliance documentation attached to that machine is not.
| Market / Regulatory Body | Key Documentation Requirement | Common Supplier Gap | What to Specifically Request |
|---|---|---|---|
| USA / FDA (21 CFR Part 211) |
cGMP compliance statement, 21 CFR Part 11 audit trail, equipment qualification protocols | PLC audit trail not user-controllable; IQ/OQ templates not FDA-format | Request FDA-format IQ/OQ/PQ templates and a 21 CFR Part 11 gap analysis for the HMI system |
| EU / EMA (EU GMP Annex 1, 15) |
Design Qualification (DQ), IQ/OQ/PQ, equipment change control procedure | Annex 15 (2015) qualification framework not followed; change control SOP missing | Request Annex 15-compliant DQ report and a sample change control template for equipment modifications |
| WHO / PQ markets (TRS 992 Annex 3) |
WHO GMP certificate for supplier facility, equipment qualification per WHO TRS 937 | Supplier facility is not WHO GMP certified; audit evidence unavailable | Request the supplier's WHO GMP certificate or equivalent national GMP certificate with valid expiry date |
| Southeast Asia (ASEAN MRA) |
ASEAN Common Technical Dossier (ACTD) packaging section, stability data Zone IVb | Stability data generated at Zone II conditions (25°C/60% RH), not Zone IVb (30°C/75% RH) | Require Zone IVb stability test results or Zone IVb extrapolation documentation before finalizing film specification |
| Latin America (ANVISA, INVIMA, ANMAT) |
Equipment qualification dossier section, Spanish/Portuguese documentation | Documentation in English only; no translated IQ/OQ templates provided | Require documentation templates deliverable in Spanish or Portuguese, or budget for certified translation before dossier submission |
I won't tell you the regional compliance question is simple. It involves your regulatory market, your dossier timeline, your relationship with the local reviewer, and sometimes your ability to finance an 8-month delay. What I can say: every project where the regional compliance requirement was specified in the URS before supplier selection finished faster than every project where it wasn't. The correlation is 100% in my sample.

Regional regulatory requirements differ — a supplier who cannot provide market-specific documentation adds months to your dossier timeline.
The True Cost of Choosing the Wrong Blister Packaging Machine Supplier
Three weeks into a job in Jakarta in 2020, I was sitting with a production manager going through a reconciliation spreadsheet. Machine had been running for four months. The numbers looked fine until we got to the service line items. I asked him to add up the six service calls they'd made to the supplier since commissioning. $127,000. Machine purchase price: $68,000.
That's the number nobody puts in the purchase justification. The supplier wasn't incompetent — the machine ran. But the PLC was proprietary, the documentation was incomplete, and two of the six service calls were regulatory responses to audit findings that proper IQ documentation would have closed pre-installation.
| Hidden Cost Category | Typical Range | Trigger Condition |
|---|---|---|
| Dossier resubmission delay (documentation gaps) | 6–8 months lost time $200k–$500k revenue deferral |
IQ/OQ not accepted by regulatory reviewer |
| Proprietary PLC service fees | $38,000–$60,000/year | Annual service contract, sole-source only |
| Forming tooling rework post-FAT | $12,000–$35,000 + 6–10 weeks downtime | Product-specific cavity spec not tested at FAT |
| Multi-vendor integration consultancy | $40,000–$120,000 per project | Blister machine and cartoner from different suppliers with undocumented interface |
| Batch recall (packaging root cause) | $1.2M–$3.8M average | Seal integrity failure, wrong film specification, or GMP non-compliance |
| Emergency on-site service (non-contracted) | $2,400–$4,500/day + travel + 14–21 day lead time | No named engineer, no service SLA in contract |
The cost of choosing the wrong blister packaging machine supplier is not the machine price. It is the sum of documentation delays, proprietary service fees, and multi-vendor integration hours that appear in the 18 months after commissioning. In every project I've reviewed where these costs exceeded the original machine price, the shortlisting process had started with a price-first RFQ. — Forester Xiang, HIJ Machinery
FAQ: Blister Packaging Machine Supplier Evaluation
❓ What is the most important criterion when evaluating a blister packaging machine supplier?
cGMP documentation completeness is the single most important criterion — specifically, whether the supplier delivers IQ/OQ/PQ protocol templates before installation, not after. Without this documentation, your equipment qualification cannot be completed, and your regulatory dossier cannot be submitted. A machine that runs but cannot be validated is not a functional asset in a licensed pharmaceutical facility.
❓ How do I verify a supplier is genuinely GMP-compliant, not just claiming compliance?
Request the supplier's manufacturing facility GMP certificate — issued by their national competent authority or a recognized body such as WHO. Then request a sample IQ/OQ protocol from a reference installation completed in the past two years. Call the reference site QA manager directly and ask whether the qualification was accepted by the regulatory reviewer on first submission. That answer is more informative than any certificate on a brochure.
❓ Is a video-call FAT acceptable for pharmaceutical blister packaging equipment?
No. A video-call FAT is not acceptable for pharmaceutical-grade equipment destined for a regulated market. It proves the machine powers on. It does not prove cavity depth consistency, sealing integrity at your film specification, product integrity at your speed, or format changeover time. All four of those parameters have caused post-installation failures in projects I've reviewed. A witnessed FAT with your product or a qualified surrogate, at the supplier's facility, is the minimum standard.
❓ Why does PLC openness matter for a blister packaging machine purchase?
A proprietary PLC locks you into sole-source service. In Southeast Asia and Latin America, sole-source service from a Chinese manufacturer typically costs $2,400–$4,500 per day on-site, with 14–21 day lead times. An open-architecture PLC (Siemens, Allen-Bradley, Omron) gives you access to a global support network at $80–$150/hr, remote resolution capability, and 21 CFR Part 11-compliant audit trails you control independently.
❓ How many suppliers should I shortlist before deciding?
Three is the standard number I recommend — enough for price competition, not so many that evaluation becomes unfocused. More important than the number is the qualification gate: only suppliers who pass the binary criteria (complete documentation package and willingness to do a witnessed FAT with your product) should make the shortlist. In my experience, that gate eliminates 40–60% of RFQ respondents before price is discussed.
❓ What should I look for in a blister packaging machine supplier's after-sales support structure?
Four specific items: a named engineer (not a department), a written response SLA (2 hours remote, 72 hours on-site decision), remote PLC access capability, and a regional service presence within 4 hours flight time of your facility. The most common failure mode I see is a supplier with excellent sales support and no post-commissioning service infrastructure. Ask for all four items in writing before you sign. If the supplier hesitates, that hesitation is your answer.
❓ Does buying a turnkey blister line from one supplier actually reduce risk?
Yes — if and only if that supplier contractually owns the interface between machines, not just each machine individually. The risk in multi-supplier lines is not machine quality; it is interface accountability. When a seal integrity failure occurs at month 8, the blame loop between blister supplier, film supplier, and cartoner manufacturer can last 3–6 months. A single supplier accountable for the complete line eliminates that loop. For a deeper look at what a complete line includes, see our guide on what is included in a turnkey pill blister packaging line.
❓ What is a reasonable lead time from order to commissioning for a pharmaceutical blister packaging machine?
For a standard thermoforming blister machine at 80–200 blisters/min, expect 12–18 weeks from deposit to FAT-ready. Cold-form Alu-Alu machines typically run 16–22 weeks due to forming station complexity. Add 3–6 weeks for shipping and 4–8 weeks for IQ/OQ/PQ after arrival. Total project timeline to production-ready: typically 8–12 months from contract signature, depending on your regulatory market and internal approval process.
📦 Ready to Evaluate Your Next Blister Line Supplier?
Get a Structured Supplier Evaluation — Not Just a Quote
HIJ Machinery supplies IQ/OQ/PQ documentation, witnessed FAT at our Wenzhou facility, open Siemens PLC architecture, and a named after-sales engineer for every project. One accountable partner from URS to compliance sign-off. Tell us your product, your target market, and your output requirement — we'll respond with a technical compliance matrix, not a catalog page.
Related reading: DPP-260 Pill Blister Packaging Machine | How to Choose a Pill Blister Packaging Machine | Pill Blister Packaging Machine Cost Breakdown