📦 Complete Guide — Syringe Blister Pack
What Is a Syringe Blister Pack?
The format behind hundreds of millions of safely delivered injections every year — and the one packaging decision that catches more pharma teams off guard than any other.
Most packaging teams encounter syringe blister packs for the first time as a finished-product specification handed down from a regulatory dossier. The format looks simple. It is not. Behind the cavity, the lidding, and the peel seal lies a sterile barrier system governed by ISO 11607, validated under ICH Q1A(R2) stability protocols, and increasingly required by procurement teams across Southeast Asia, the Middle East, and Latin America for any injectable product targeting hospital or pharmacy channels.
This guide covers every layer of what a syringe blister pack actually is — materials, forming methods, compliance requirements, and the machine formats used to produce them at commercial scale. If you are preparing a URS, evaluating a line, or simply trying to explain the format to a new team member, this is the reference you need.
What Exactly Is a Syringe Blister Pack and Why Does the Definition Matter?
🎯 Direct Answer
A syringe blister pack is a single-unit sterile barrier packaging system in which a thermoformed or cold-formed plastic or aluminum cavity holds one pre-filled syringe, sealed by a gas-permeable or impermeable lidding material — maintaining container closure integrity for a validated shelf life of 24 to 36 months.
The word “blister” is used loosely across industries. For medical device packaging — and a pre-filled syringe is classified as a combination product in most jurisdictions — the definition carries regulatory weight. ISO 11607-1:2019 defines a sterile barrier system as the “minimum packaging that minimizes the risk of ingress of microorganisms and allows aseptic presentation of the product at the point of use.” A syringe blister pack, when correctly specified and validated, is exactly that.
The format became the dominant individual-dose presentation for pre-filled syringes in regulated markets during the 2000s, displacing bulk trays for most retail and hospital pharmacy channels. Three drivers made it standard: individual dose accountability, needle-stick risk reduction at the point of care, and compatibility with unit-dose serialization systems required under the EU Falsified Medicines Directive and US DSCSA.
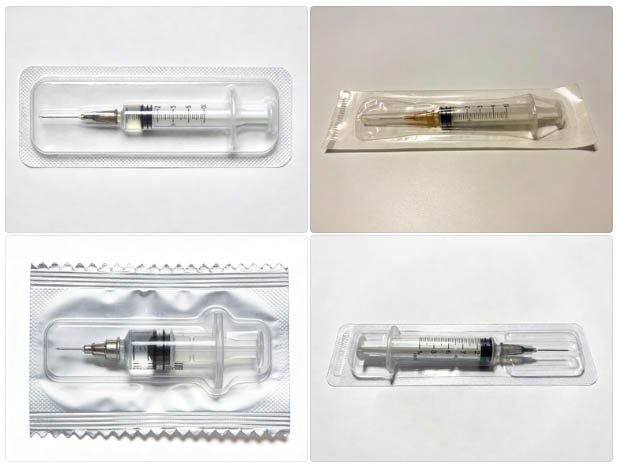
A pre-filled syringe individually sealed in a thermoformed cavity — the format that defines individual-dose sterile packaging for injectable products.
What Are the Two Main Types of Syringe Blister Pack Forming Methods?
🎯 Direct Answer
Syringe blister packs are produced by one of two forming methods: thermoforming, which heat-forms PVC, PETG, or PP film into cavities; or cold forming (Alu-Alu), which mechanically presses aluminum laminate into cavities at ambient temperature — the second method offering near-zero moisture vapor transmission for moisture-sensitive drug products.
The choice between thermoforming and cold forming is not aesthetic. It determines your moisture barrier, your tooling cost, your cycle speed, and — in markets like tropical Southeast Asia or sub-Saharan Africa — whether your product survives distribution at all.
Thermoformed Syringe Blister Packs
Thermoforming heats a plastic web film to its softening point, then draws or plugs it into a cavity mold. For syringe blister packs, the cavity geometry is three-dimensional and asymmetric — accommodating the barrel body, flange, and needle shield simultaneously. Standard forming materials include rigid PVC (250–350 μm), PETG, and polypropylene. Moisture vapor transmission rates (MVTR) for PVC at 250 μm run 4–6 g/m²/day. Adequate for most stable API profiles; not adequate for moisture-sensitive biologics or lyophilized formulations.
Thermoforming is faster — typical cycle speeds of 60–120 blisters per minute on a mid-range pharmaceutical blister line — and tooling investment is lower than cold form tooling. That combination makes it the default for standard injectable products destined for temperate-climate markets.
Cold-Formed (Alu-Alu) Syringe Blister Packs
Cold forming presses an aluminum laminate — typically OPA/Alu/PVC or OPA/Alu/PE construction — into the cavity geometry at room temperature using mechanical forming plugs. The result is an opaque, metallic cavity with MVTR values below 0.5 g/m²/day. No heat. No optical clarity. Total moisture barrier. For pre-filled syringes containing hygroscopic API formulations or photosensitive drug substances, cold-formed Alu-Alu packaging is not a premium option — it is a regulatory requirement in Zone IVb markets.
| Forming Method | Primary Material | MVTR (g/m²/day) | Typical Cycle Speed | Best For | Key Limitation |
|---|---|---|---|---|---|
| Thermoforming | PVC / PETG / PP | 4–6 (PVC 250μm) | 60–120 blisters/min | Stable APIs, temperate markets | Limited moisture protection for Zone IVb |
| Cold Forming (Alu-Alu) | OPA / Alu / PVC laminate | <0.5 (typical 0.02–0.5) | 30–80 blisters/min | Moisture-sensitive, tropical markets | Higher tooling cost; opaque cavity |
| Thermoforming + PVDC | PVC/PVDC co-extrusion | 0.8–1.5 | 60–110 blisters/min | Mid-barrier applications | PVDC disposal restrictions in EU markets |
⚠️ Field Note — Forester Xiang, HIJ Machinery
I saw this failure pattern in a plant outside Kuala Lumpur in 2020. The stability data had been generated in Europe at 25°C/60% RH. The product was bound for peninsular Malaysia — Zone IVb: 30°C/75% RH. The team had specified thermoformed PVC because that was what their European contract packer used. Nobody flagged the MVTR gap until the 6-month accelerated data came back with moisture uptake exceedances. The blister line was already ordered. Retooling for cold-form Alu-Alu cavities added fourteen weeks and cost more than the original tooling budget. The lesson is not subtle: specify your forming method from your ICH Q1A(R2) target market conditions, not from your reference site’s historical practice.
What Materials Make Up a Syringe Blister Pack? (Cavity + Lidding Explained)
🎯 Direct Answer
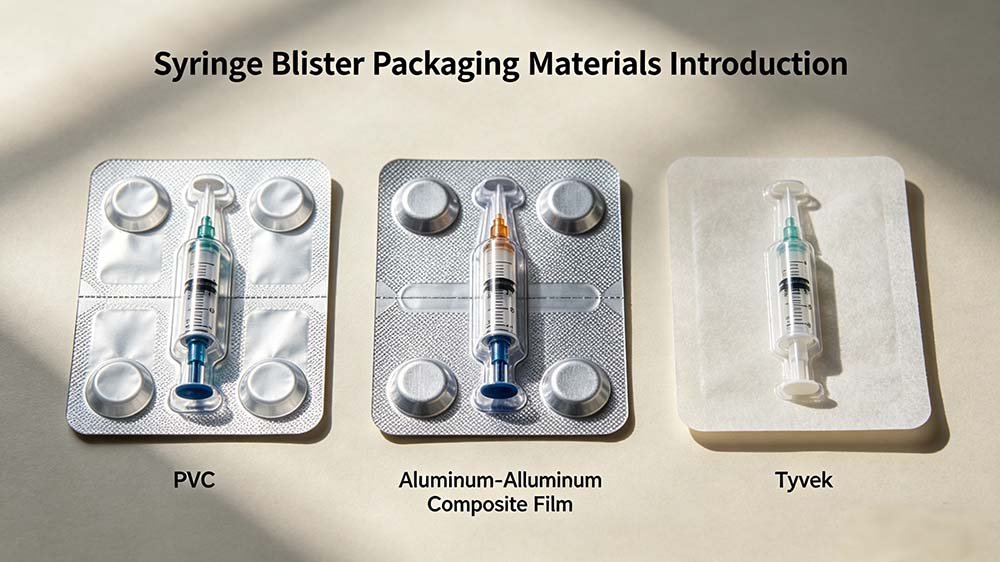
A syringe blister pack consists of two functional layers: a formed cavity (PVC, PETG, PP, or aluminum laminate) that holds the syringe, and a lidding material — most commonly medical-grade Tyvek® (spunbond HDPE) for thermoformed packs or aluminum foil for cold-formed packs — that is heat-sealed to create the sterile barrier and allows controlled peelback at the point of use.
Material selection is where most URS documents either get specific or leave a gap that causes problems later. The cavity and the lidding must be validated together as a system — not as individual components. ISO 11607-1 requires that the entire sterile barrier system be validated for seal integrity, burst resistance, and aging. Switching lidding supplier mid-lifecycle is a change control event that triggers re-validation.
Cavity Materials for Syringe Blister Packs
- Rigid PVC (250–350 μm): Most common for standard thermoformed syringe packs. Excellent formability, cost-effective, good optical clarity for visual inspection. MVTR 4–6 g/m²/day limits use in high-humidity markets.
- PETG / PET: Higher clarity than PVC, more rigid, no chlorine content. Preferred by teams targeting EU markets with PVC restrictions. Slightly higher forming temperature requirement; cavity definition remains excellent.
- Polypropylene (PP): Chemical resistance, suitable for autoclave sterilization processes. Less common for syringe blister packs but used in specific orthopedic device presentations.
- OPA/Alu/PVC laminate (Alu-Alu): Cold-form construction. Aluminum core provides total moisture and light barrier. The OPA (oriented polyamide) outer layer provides puncture resistance; the PVC inner layer provides heat-seal compatibility with aluminum lidding foil.
Lidding Materials for Syringe Blister Packs
Tyvek® (DuPont spunbond HDPE) is the dominant lidding material for thermoformed syringe blister packs in regulated pharmaceutical markets. It is gas-permeable — essential for EO (ethylene oxide) sterilization processes — while maintaining microbial barrier performance. Peel force is typically specified at 2–8 N/15 mm depending on the application, ensuring clean peel at the point of use without the lidding tearing or delaminating and contaminating the sterile field.
For cold-formed Alu-Alu syringe blister packs, the lidding is aluminum foil laminate — typically Alu/PVC or Alu/PE construction. These systems are not EO-compatible; terminal sterilization is achieved by gamma or e-beam irradiation applied post-sealing. The hermetic seal provides the same barrier performance as the cold-formed cavity.

HIJ Machinery syringe blister packing machine at commercial-scale production — thermoforming PVC cavities with Tyvek lidding for pre-filled syringe packaging.
Why Is Blister Packaging Used for Syringes Instead of Bulk Tray or Pouch Formats?
🎯 Direct Answer
Blister packaging is used for pre-filled syringes because it delivers individual-unit sterile barrier protection, prevents needle-stick injury during product handling, supports unit-level serialization required under FDA DSCSA and EU FMD, and reduces contamination risk compared to multi-unit tray or pouch formats used in bulk hospital supply chains.
The bulk tray format — multiple syringes nested in a thermoformed tray under a single Tyvek lid — still exists in hospital pharmacy repacking workflows and clinical trial supply chains. It is not the retail pharmacy or point-of-care format. Individual blister packs became standard for several converging reasons.
880,000
Needle-stick injuries reported annually in U.S. healthcare settings (CDC/EPINet data)
ISO 11607
Global standard governing sterile barrier system design and validation for medical device packaging
24–36 mo
Typical validated shelf life for syringe blister packs under WHO GMP storage conditions
EU MDR
EU Medical Device Regulation mandating unit-level unique device identification (UDI) on combination products
The needle-stick figures are not abstract. In West African hospital networks I visited in 2022, repacking staff handling bulk-tray syringes without individual protection reported injury rates that local procurement teams described as “accepted” as part of the workflow. They were not accepted — they were untracked. Individual blister packaging forces a design barrier between the needle and unprotected hands at every point from warehouse to bedside. That is not a marketing claim. It is an engineering constraint with a measurable outcome.
Serialization is the second driver. Unit-level barcodes or 2D DataMatrix codes — required under EU FMD for prescription medicines and under FDA DSCSA for prescription drug products — require a discrete packaging unit for each code. A bulk tray gives you one serial number for 10 syringes. A blister pack gives you one serial number per syringe. Regulators in Brazil (ANVISA RDC 330), India (CDSCO), and Saudi Arabia (SFDA) have progressively aligned with the unit-level traceability requirement. Individual blister packaging is the practical substrate for compliance.
Of 47 emerging-market injectable packaging projects I have managed since 2015, 31 required individual syringe blister packs specifically to meet local serialization or needle-stick prevention regulatory requirements — not because the client preferred the format, but because their target market required it. — Forester Xiang, HIJ Machinery
How Is a Syringe Blister Pack Produced? The Machine Process Step by Step
🎯 Direct Answer
A syringe blister pack is produced in six sequential machine stations: film heating and forming (thermoform) or plug-assisted cold forming (Alu-Alu), syringe loading (manual or automated), lidding material application, heat-seal station, die-cutting, and finished-pack discharge — with optional inline vision inspection and serialization printing between the sealing and discharge stations.
- 1 Film Loading & Cavity Forming The forming web film (PVC roll or Alu-Alu laminate) is fed from a dual-roll holder. In thermoforming, the film passes through a heating plate station where it is softened to 120–180°C, then drawn into the cavity mold by vacuum and/or compressed air. Cold-form Alu-Alu uses mechanical forming plugs at ambient temperature. Cavity dimensions — critical for syringe flange clearance and needle shield height — are defined by the mold tooling.
- 2 Syringe Loading Pre-filled syringes are loaded into the formed cavities, either manually by operators (common on low-speed lines) or via automated syringe feeding systems using pick-and-place robots or vibratory/slide-in feed mechanisms. Loading orientation — needle end forward or barrel-first — is machine-configuration dependent and must be specified in the URS.
- 3 Lidding Application The lidding web (Tyvek or aluminum foil) unwinds from the upper roll and is indexed over the loaded cavities. Precise web tension control is required to prevent lidding distortion, which directly affects seal integrity at the subsequent heat-seal station.
- 4 Heat Sealing The sealing station applies controlled temperature (typically 150–200°C for Tyvek/PVC interfaces), pressure (2–5 bar), and dwell time to create the peel seal. These three parameters are the IQ/OQ validation critical parameters. The seal must pass ASTM F88 peel strength testing and USP <1207> container closure integrity evaluation as part of the validation package.
- 5 Die Cutting & Separation The sealed web is die-cut to individual blister card dimensions. Rotary or flat-bed punch tooling defines card geometry. Scrap web is wound onto a waste reel or granulated inline.
- 6 Discharge, Inspection & Serialization Finished blister cards are discharged to a conveyor leading to optional vision inspection systems (checking seal completeness, syringe presence, and label registration) and inkjet or laser serialization printing. Cards are then conveyed to a downstream cartoning machine for secondary packaging.

Syringe blister packing machine application — the full forming-loading-sealing-cutting sequence that produces validated sterile blister packs at commercial scale.
📋 Applicable Regulatory Standards — Syringe Blister Pack Validation
What Compliance Standards Apply to Syringe Blister Packs in Regulated Markets?
🎯 Direct Answer
Syringe blister packs must comply with ISO 11607 (sterile barrier system design and process validation), USP <1207> (container closure integrity), ASTM F88 (seal strength), and — depending on target market — EU MDR 2017/745, FDA 21 CFR Part 211, or WHO GMP TRS 992 Annex 3, with aging validation conducted per ICH Q1A(R2) using real-time and accelerated stability protocols.
The compliance chain for syringe blister packs is longer than most packaging teams expect the first time they build it. ISO 11607 alone has two parts: Part 1 covers material requirements and sterile barrier system design, while Part 2 governs process validation for forming, sealing, and assembly. Both are required. The common gap I see in emerging-market submissions is a complete Part 1 technical file with no Part 2 process validation records — valid material qualification; zero evidence that the sealing process is controlled.
The CDSCO reviewer in a 2021 submission I supported cited exactly this gap. The dossier contained material certificates, accelerated stability data, and supplier declarations for the Tyvek lidding. It did not contain sealing process validation records (temperature, pressure, dwell time qualification with statistical process capability data). The file went back. Four months of review cycle burned. The seal parameters had been validated — they just had not been documented in the submission format the reviewer expected.
For WHO prequalification submissions, the same gap recurs with regularity. WHO TRS 992 Annex 3 requires that “critical packaging operations shall be validated, documented, and the validated state maintained.” That sentence, 14 words, covers a validation package that routinely runs to 80–120 pages. If your machine vendor cannot supply IQ/OQ execution records for the specific sealing station configuration on your line, you are building that documentation yourself. That is a project risk worth pricing before you sign the purchase order.
⚠️ Diagnostic: Syringe Blister Pack Compliance Gaps
🔴 Symptom: Regulatory submission returned for packaging section
Likely cause: ISO 11607-2 process validation records absent or incomplete — material qualification present but sealing parameter qualification missing.
✅ Fix: Prepare a dedicated sealing validation report documenting temperature/pressure/dwell time qualification with Cpk ≥1.33 for all three parameters. Include challenge samples at specification limits.
🔴 Symptom: Blister pack fails 6-month accelerated stability (moisture uptake exceedance)
Likely cause: Forming material MVTR insufficient for target market climate zone — thermoformed PVC specified for Zone IVb distribution.
✅ Fix: Upgrade to cold-form Alu-Alu cavity construction or PVC/PVDC co-extrusion. Re-run accelerated stability at 40°C/75% RH per ICH Q1A(R2) Zone IVb conditions before market launch.
🔴 Symptom: Seal integrity failure during CCI testing (dye ingress at <60 mbar)
Likely cause: Heat-seal parameter drift during production, or lidding web tension inconsistency causing incomplete bonding at cavity edge radius.
✅ Fix: Audit sealing station temperature uniformity across the full seal platen. Check lidding web tension control calibration. Run ASTM F2338 dye-ingress CCI on 30-unit sample across three production batches to establish process capability baseline.
What Types of Machines Produce Syringe Blister Packs at Pharmaceutical Scale?
🎯 Direct Answer
Syringe blister packs are produced on dedicated syringe blister packing machines — either thermoforming or cold-form configured — running at 30–150 blisters per minute depending on the model, with servo-driven forming, sealing, and cutting stations, and optional integration with automated syringe feeding, serialization printing, and downstream cartoning equipment.
Not all blister packing machines handle syringes. Tablet and capsule blister machines run flat, shallow cavities with simple geometry. Syringe blister machines require deep, three-dimensional cavity forming — typically 40–80 mm cavity depth for standard 1 mL to 5 mL pre-filled syringes — and a syringe loading mechanism capable of handling a fragile glass or plastic barrel without stress fracture. That combination narrows the equipment field considerably.
At HIJ, the machines configured for syringe blister packing use servo-motor-driven forming and sealing stations with PLC-controlled parameter management. The forming mold is syringe-specific: cavity geometry is matched to flange diameter, needle shield height, and barrel length from the URS. I have seen generic machine vendors quote “syringe-compatible” blister machines with standard tablet molds and a verbal assurance that the mold can be adapted. That conversation usually ends at FAT when the needle shield does not clear the cavity edge on the first production trial.
| Machine Configuration | Speed Range | Forming Method | Syringe Size Range | Automation Level | Typical Application |
|---|---|---|---|---|---|
| Semi-Automatic Syringe Blister | 10–30 blisters/min | Thermoforming | 1–5 mL | Manual loading; auto seal & cut | Clinical trial, low-volume launch |
| Automatic Thermoforming | 40–120 blisters/min | Thermoforming PVC/PETG | 0.5–10 mL | Automated feed & vision inspection | Commercial pharma, hospital supply |
| Automatic Cold-Form (Alu-Alu) | 30–80 blisters/min | Cold forming Alu laminate | 1–10 mL | Automated feed & serialization | Moisture-sensitive APIs, Zone IVb |
| Turnkey Integrated Line | Up to 150 blisters/min | Thermoform or cold-form | 0.5–20 mL | Full automation: fill → blister → carton | High-volume commercial production |
For teams evaluating machine options, the decision tree starts with output volume and syringe specification — not with price. I will address that selection framework in detail in our guide on how to choose the right syringe blister packaging machine for your pharma line. The short version: speed requirements drive capital cost far more than forming method, and the syringe specification determines tooling cost before any machine quotation is meaningful.
How Is a Syringe Blister Pack System Validated Before Commercial Production?
🎯 Direct Answer
Syringe blister pack validation follows a DQ/IQ/OQ/PQ sequence: Design Qualification confirms the system meets URS requirements; Installation Qualification verifies correct installation; Operational Qualification establishes and qualifies the critical process parameters; and Process Qualification demonstrates reproducible output under production conditions — supported by packaging aging and accelerated stability studies per ICH Q1A(R2).
Validation is not an afterthought. It is the proof that your blister pack reliably does what it is claimed to do throughout its shelf life. For syringe blister packs specifically, three validation dimensions run in parallel: machine process validation (sealing parameters), sterile barrier system validation (ISO 11607 compliance), and container closure integrity (USP <1207> testing). All three must be completed and documented before commercial release.
The OQ stage is where most teams encounter their first hard constraint. OQ requires establishing the operating ranges for critical parameters — sealing temperature ±5°C, sealing pressure ±0.2 bar, dwell time ±0.1 sec — and demonstrating that packs produced at all combinations of these range limits pass seal integrity, burst, and peel strength specifications. That requires a designed experiment, not a single-point “golden batch.” Teams that skip the range qualification and run only at the target setpoint create a silent vulnerability: any process drift toward the range edge triggers non-compliance without any prior evidence that the edge is safe.
I have seen this exact situation in a plant in São Paulo. The sealing temperature drifted 8°C over four months due to thermocouple calibration drift. The OQ had been done at a single setpoint. No range qualification. The annual stability check found delamination at the peel test. By then, 18 batches had been released. The recall cost was not the machine price. It was the 18 batches.
📌 GMP Compliance Note
For teams preparing validation documentation for WHO prequalification or CDSCO submissions, the complete GMP compliance and validation requirements guide for syringe blister packaging covers the full DQ through PQ documentation structure, including the specific IQ/OQ protocol content that regulatory reviewers have requested in Southeast Asian and African market submissions. EU GMP Annex 15 (2015) and WHO TRS 992 Annex 3 are the applicable validation frameworks for most regulated market submissions.
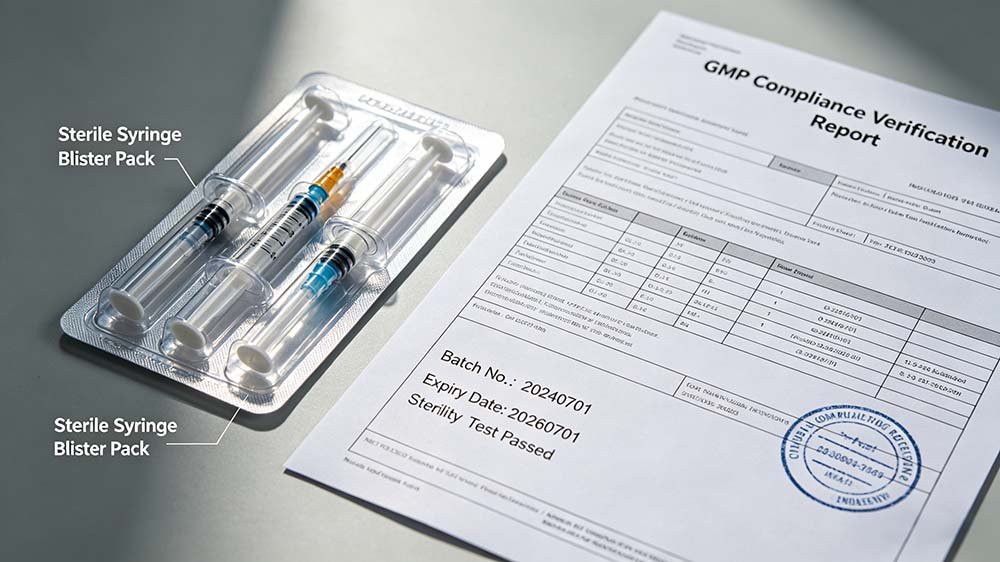
GMP validation process for syringe blister packing lines — IQ/OQ/PQ execution with documented sealing parameter qualification per EU GMP Annex 15 and WHO TRS 992.
Syringe Blister Pack vs. Tray Pack vs. Pouch: Which Format Is Right for Your Product?
🎯 Direct Answer
Individual syringe blister packs are the preferred format for retail pharmacy and point-of-care distribution where unit-level serialization, needle-stick prevention, and individual dose accountability are required. Tray packs suit bulk hospital procurement and clinical trials. Pouches are used for rigid glass syringe formats where cavity depth would be impractical. Most commercial pharmaceutical market launches targeting regulated retail channels require individual blister packaging.
The format decision is not one-size-fits-all, and it is made correctly by mapping distribution channel requirements before touching a URS. Three variables dominate the decision: regulatory serialization requirement at the destination market, syringe geometry (glass vs. plastic barrel, needle shield type, flange design), and required moisture barrier performance for the target climate zone.
| Packaging Format | Serialization Support | Needle-Stick Protection | Moisture Barrier | Unit Cost Index | Best Market Channel |
|---|---|---|---|---|---|
| Individual Blister Pack | ✅ Unit-level 2D code | ✅ Rigid cavity barrier | Medium–High (material-dependent) | 1.0 (reference) | Retail pharmacy, hospital dispensing |
| Multi-Syringe Tray Pack | ⚠️ Pack-level only | ⚠️ Shared lid, open on breach | Medium (Tyvek lid) | 0.6–0.7 | Hospital pharmacy bulk, clinical trial |
| Peel Pouch (Chevron/Header) | ⚠️ Pouch-level only | ⚠️ Soft format, less rigid barrier | Low–Medium | 0.5–0.6 | Glass syringe formats, OEM supply |
| Cold-Form Alu-Alu Blister | ✅ Unit-level 2D code | ✅ Rigid cavity barrier | ✅ Very High (<0.5 g/m²/day) | 1.3–1.6 | Tropical markets, moisture-sensitive API |
The cost differential between individual blister and tray pack formats is real. I have sat across from procurement directors who showed me the numbers: tray pack at 40% lower per-unit packaging cost for a 2-million-unit annual volume. That is a significant saving. The question I ask is whether the target market pharmacy channel accepts tray format for over-the-counter injectable products. In most Southeast Asian and African pharmacy retail contexts, the answer is no. The individual blister pack is the only format that moves through the retail channel. Tray packs require repacking at the pharmacy level, which reintroduces contamination risk and eliminates the serialization advantage.
Frequently Asked Questions: Syringe Blister Pack
What is a syringe blister pack?
A syringe blister pack is a sterile barrier packaging system consisting of a thermoformed or cold-formed cavity that individually holds one pre-filled syringe, sealed with Tyvek or aluminum foil lidding — maintaining container closure integrity and sterility protection throughout a validated shelf life of 24 to 36 months under ICH Q1A(R2) storage conditions.
What material is used for syringe blister packs?
The forming cavity is typically rigid PVC (250–350 μm), PETG, or cold-form OPA/Alu/PVC laminate. The lidding is medical-grade Tyvek® (spunbond HDPE) for thermoformed packs permitting EO sterilization, or aluminum foil laminate for cold-formed packs. Material selection is driven by the product’s moisture sensitivity, target market climate zone, and sterilization method. For moisture-sensitive APIs distributed in tropical markets (ICH Zone IVb: 30°C/75% RH), cold-form Alu-Alu construction reduces MVTR to below 0.5 g/m²/day compared to 4–6 g/m²/day for standard PVC — a 10x or greater reduction depending on gauge.
Does a syringe blister pack need to comply with ISO 11607?
Yes. ISO 11607 is the primary international standard for sterile medical device packaging, including pre-filled syringe blister packs classified as combination products. Both Part 1 (material and design requirements) and Part 2 (process validation) are required for regulatory submissions in EU, US FDA, and WHO prequalification dossiers.
What machine is used to make syringe blister packs?
Syringe blister packs are produced on dedicated syringe blister packing machines configured with deep three-dimensional forming molds sized to the syringe specification (barrel length, flange diameter, needle shield height). Standard commercial lines run at 40–120 blisters per minute. Cold-form Alu-Alu configurations run at 30–80 blisters per minute due to the mechanical forming cycle. Machine selection must be matched to annual output volume, syringe geometry, and GMP validation requirements before issuing a purchase order.
How long does syringe blister pack validation take?
Full DQ/IQ/OQ/PQ validation for a syringe blister packing machine and its sterile barrier system typically takes 4–8 months from equipment installation to final PQ report sign-off, depending on whether real-time aging studies are required to supplement accelerated stability data. Accelerated stability alone (6 months at 40°C/75% RH) adds 6 months to the timeline. Teams targeting WHO prequalification or CDSCO submission should plan for 12–18 months from URS to validated status. That is not the machine vendor’s lead time — it is the regulatory evidence-building period.
Can the same blister machine handle both syringes and tablets?
Not typically on a single mold configuration. Syringe cavities require 40–80 mm forming depth and three-dimensional geometry; tablet cavities are 3–8 mm deep. Some machines are designed for mold changeover flexibility, but the forming station depth range and the product loading mechanism are fundamentally different. Dual-product machines exist for R&D or multi-product facilities but carry higher tooling inventory cost and longer changeover validation requirements.
Is Tyvek or aluminum foil better as lidding for syringe blister packs?
Tyvek is better for products requiring EO sterilization post-packaging, because it is gas-permeable. Aluminum foil provides a hermetic seal with superior moisture and light barrier — essential for moisture-sensitive formulations and photosensitive drug substances. The choice is determined by your sterilization method and your ICH stability target market, not by cost.

Completed syringe blister pack output from a GMP pharma blister packaging line — individual sterile barrier packs ready for secondary cartoning and serialization.
The Bottom Line on Syringe Blister Packs: What Every Pharma Packaging Team Needs Before Specifying Equipment
A syringe blister pack is not a commodity packaging decision. The format, the material, the lidding, the forming method, and the machine that produces it are all interconnected with your product’s regulatory dossier, stability profile, and target market distribution chain. Getting any one element wrong — the MVTR specification for a tropical market, the cavity geometry for a new syringe supplier’s barrel, the seal parameter validation scope for a WHO submission — costs time that cannot be recovered before a market launch deadline.
The teams that navigate this format successfully share one habit: they start the packaging specification at the same time as the formulation stability study, not after it. They know their target climate zone before they know their syringe blister pack machine model number. They have a validated URS with real syringe components before they accept FAT at the vendor’s facility.
If you are at the beginning of that process — or trying to recover from a specification that started in the wrong order — the next logical step is understanding exactly what to look for when choosing the right machine configuration for your line. That is covered in detail in our guide on how to choose the right syringe blister packaging machine for your pharma line.
For the full compliance and validation framework — DQ through PQ, ISO 11607, and what CDSCO and WHO reviewers specifically ask for — the GMP compliance and validation requirements guide for syringe blister packaging covers the documentation structure in full. And if you want to see the full machine specification for HIJ’s syringe blister packing line, the syringe blister packing machine product page contains the current technical specifications and configuration options.
The syringe blister pack decision cannot be unmade at FAT. By the time a machine is on your floor, your forming method, your cavity geometry, and your lidding system are committed. The validation timeline is set. The only moment to get it right is before the purchase order — and that moment is shorter than most teams think. — Forester Xiang, Founder, HIJ Machinery
Ready to Specify Your Syringe Blister Pack Line?
HIJ Machinery engineers syringe blister packing machines around your exact syringe specification — barrel dimensions, flange geometry, needle shield height — with full IQ/OQ/PQ documentation support and co-engineering from URS to FAT. Get a line configuration matched to your product and your market.