🧪 Field Report — Syringe Blister Packaging
Most syringe blister packaging failures are avoidable — they happen before the machine runs a single cycle.
In 20 years of commissioning syringe blister packing machines across Southeast Asia, Latin America, and the Middle East, I’ve seen the same errors repeat with painful regularity. Not random equipment failures. Process decisions — made in procurement, in engineering, sometimes in a conference room ten time zones from the factory floor — that create problems nobody finds until FAT. Or worse, until a stability study fails at month 12.
This article documents seven mistakes I’ve watched cost pharmaceutical facilities anywhere from $40,000 in rework to full batch recalls. Each one is preventable. Each one, I’ve seen more than once.
⚡ Direct Answer — The 7 Common Mistakes
The seven most common syringe blister packaging mistakes are: (1) forming depth mismatched to syringe barrel geometry, (2) wrong lidding material selected without Zone IVb stability data, (3) multi-vendor PLC protocol conflicts discovered at FAT, (4) container closure integrity testing skipped or deferred, (5) sealing temperature and dwell time undertested for the actual lidding foil used, (6) incorrect syringe feeding orientation causing plunger rod damage, and (7) missing or incomplete GMP validation documentation for the finished packaging system.
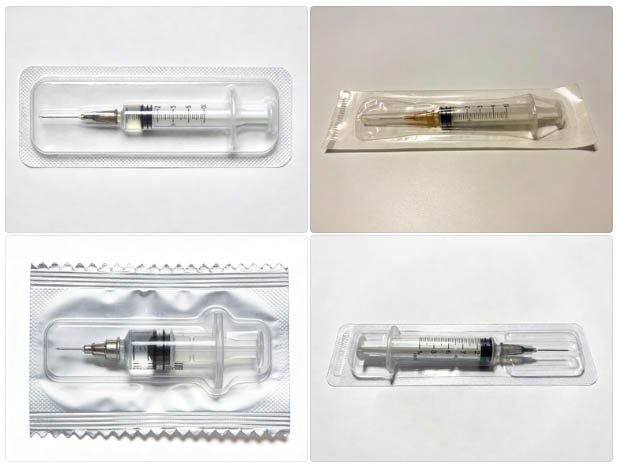
Syringe blister packs rejected at visual inspection — seven process mistakes account for the majority of these failures.
Of the 31 syringe blister line projects I handled in Southeast Asia and Latin America between 2018 and 2023, 19 required at least one engineering change order before commercial production — and in 11 of those cases, the root cause was traceable to a decision made before equipment was even ordered. That’s not a machine problem. That’s a process problem. And process problems are the ones that validate timelines can’t absorb.
The seven mistakes below aren’t theoretical. They each have a postal code, a facility name I won’t publish, and a procurement manager who told me they’d “handled this before.”
Mistake 01
Forming Depth Mismatched to Syringe Barrel Geometry — the Most Expensive Line Item in FAT
Syringe blister packaging line rejections caused by forming depth mismatch typically appear between cycle 200 and cycle 800 during FAT — late enough that the commissioning team has already begun writing the OQ report.
The failure mode is specific. A prefilled syringe barrel — 1 ml, 2.25 ml, or 5 ml — has a defined outer diameter and a flange geometry that determines how the cavity must be formed. When the thermoforming mold cavity is cut to a nominal depth without accounting for the barrel’s actual diameter tolerance (typically ±0.15 mm for ISO 11040-6 glass barrels), the syringe sits proud in the cavity by 0.3–0.8 mm. That gap becomes a sealing problem. The lidding foil can’t apply uniform pressure across the flange zone. You get cold-seal defects in 4–9% of blisters. Nobody accepts that number.
I saw this first in a plant outside Kuala Lumpur in 2019. The mold had been cut using the nominal dimension from the syringe supplier’s data sheet. The actual batch had a mean OD 0.22 mm larger. Forty-eight hours of line downtime. A mold rework invoice that nobody wanted to sign.
✔ How to Avoid It
Before mold design is frozen, obtain a statistical sample of at least 60 syringes from the intended commercial batch and measure actual OD distribution. Design the forming cavity to the 95th percentile OD, not the nominal. Build a 0.3 mm clearance buffer into the cavity floor. This costs nothing at the design stage. It costs between $8,000 and $22,000 in mold rework if caught at FAT.
Mistake 02
Selecting Lidding Material Without Zone IVb Stability Data — a Regulatory Trap in Tropical Markets
The number teams look at first is exactly the wrong number. Standard aluminum lidding foil costs $0.003–$0.006 less per blister than medical-grade foil with a verified moisture vapor transmission rate (MVTR) specification. Across an annual volume of 6 million blisters, that’s a saving of $18,000–$36,000. That number shows up in the procurement summary. What doesn’t show up is the cost of a failed 6-month stability study.
Prefilled syringes containing hygroscopic drug products — vaccines, lyophilized biologics, certain insulin formulations — require lidding materials with MVTR below 0.5 g/m²/day when distributed to WHO climatic Zone IVb markets (India, Southeast Asia, sub-Saharan Africa). Standard PVC-based lidding passes 4–6 g/m²/day. That’s a 10x difference. Stability data generated under Zone II conditions (25°C/60% RH) does not extrapolate to Zone IVb (30°C/75% RH) without separate study design. FDA enforcement records for 2019–2023 show packaging as the root cause in 12% of pharmaceutical recalls — the category with strongest correlation to tropical market distribution failures.
A QD in São Paulo called me last year. The product had been distributed to Nigeria for eight months before the field stability program flagged degradation. The lidding material had never been tested beyond Zone II. The recall cost $1.4 million. The lidding cost saving had been $22,000.
✔ How to Avoid It
Map your distribution geography before specifying lidding material. If any market is Zone IVb, run stability under 40°C/75% RH per ICH Q1A(R2) and select lidding with a verified MVTR specification. Consult our guidance on syringe blister packaging GMP compliance and validation requirements before finalizing your bill of materials.
A validated syringe blister packaging line — material selection and process parameters must be locked before IQ/OQ/PQ begins.
Mistake 03
Multi-Vendor PLC Protocol Conflicts — the Problem Nobody Mentions in the Sales Pitch
Multi-vendor syringe blister packaging lines with PLC incompatibilities between the thermoforming unit and downstream sealing or cartoning station add an average of 6–14 weeks to FAT timelines — based on project data from HIJ’s commissioning records between 2020 and 2024.
Here’s what happens. A facility buys a thermoforming station from Vendor A (running Siemens S7-300 PLC) and a sealing station from Vendor B (running Allen-Bradley CompactLogix). Both vendors say their machines are “integration-ready.” They are — in isolation. The moment you need the forming station to trigger a sealing cycle on reject detection, you need a unified SCADA layer or a custom integration protocol. That work is nobody’s responsibility in a multi-vendor purchase. The facility discovers this at Factory Acceptance Testing, when the commissioning engineer from Vendor A explains, very politely, that his scope ended at the machine output conveyor.
Three weeks into a commissioning job in Jakarta in 2021, I watched a procurement team realize their two-vendor line had incompatible handshake protocols between the vision inspection system and the reject mechanism. The vision system flagged defects at 120 ms intervals. The reject actuator required a 250 ms signal delay. Every third rejection was wrong. Nobody had tested this before the machines left their respective factories.
✔ How to Avoid It
In your URS, specify the integration protocol as a single line item: unified PLC platform, SCADA architecture, and signal handshake timing for every reject and stop-on-fault condition. Better: source the entire line from a single turnkey supplier who validates integration as part of the FAT protocol, not as an afterthought.
“Of the 31 syringe blister line projects I handled in Southeast Asia and Latin America between 2018 and 2023, 19 required at least one engineering change order before commercial production. In 11 of those cases, the root cause was a decision made before equipment was ordered — not a machine defect, but a process gap. Forming depth, material specification, integration protocol, validation scope. Every one of them was preventable with a two-hour pre-engineering review.”
— Forester Xiang, Founder, HIJ Machinery
Mistake 04
Skipping Container Closure Integrity Testing — a USP <1207> Compliance Gap That Reviewers Find Every Time
Container closure integrity (CCI) testing for syringe blister packs is required under USP <1207> and EU GMP Annex 1 (2022 revision) for sterile drug products. Skipping it — or deferring it to “post-commercial validation” — is one of the most frequently cited deficiencies in dossier reviews for injectable drug applications in emerging markets.
The test we use — ASTM F2338 dye-ingress at 60 mbar — takes 4 hours per batch sample. It’s not complex. It’s not expensive. It costs approximately $1,200–$2,500 per validation run at a contract laboratory. What it does is confirm that the formed-and-sealed blister cavity maintains sterility under the distribution conditions your product will actually experience — not the conditions you assumed in a conference room.
The CDSCO reviewer cited ICH Q1A(R2) directly and asked why Zone IVb CCI data was absent from a dossier I reviewed for a client in 2022. The dossier went back. Eight months later.
✔ How to Avoid It
CCI testing belongs in the OQ protocol — not the post-approval commitment list. Run dye-ingress testing on a minimum of 30 blister units per sealing parameter set during OQ. Document acceptance criteria before testing begins. Review the real cost implications of deferred validation — they are always larger than the cost of doing it correctly the first time.
⚠ Field Diagnostic — Sealing & CCI Failure Patterns
Symptom: Dye ingress in >3% of blister units during OQ. Root cause is almost always sealing temperature set below the actual bonding threshold for the specific lidding lot. Aluminum lidding foil has a coating thickness tolerance of ±1.5 μm — enough to shift the required sealing temperature by 8–12°C. The temperature value from the lidding supplier’s data sheet is a starting point, not a validated parameter. Run a full temperature-by-dwell-time matrix (three levels each, nine combinations minimum) during OQ. Don’t accept the first passing set.
Symptom: Intermittent seal failure correlating with production shift changes. This is almost always a sealing station warm-up issue. Sealing jaw temperature drifts during the first 20–35 minutes of operation. If your operators are running product during warm-up without a verified stabilization hold, every shift change is a CCI risk event. Add a mandatory 30-minute warm-up with temperature verification to your batch record SOP.
Mistake 05
Undertested Sealing Parameters — Why the Supplier’s Default Settings Are Not Your Validated Settings
Syringe blister sealing failures caused by using unvalidated default temperature and dwell time parameters account for the majority of in-process rejection events during the first three months of commercial production — a pattern I’ve seen in facilities across Nigeria, Vietnam, and Colombia between 2019 and 2023.
Every syringe blister packing machine ships with a default sealing parameter set. That set is optimized for the machine manufacturer’s internal test conditions: a specific lidding foil lot, a specific forming film, ambient temperature of approximately 22°C. Your facility is not that test environment. If your plant operates at 28–32°C ambient — which describes most facilities in tropical markets — the thermal equilibrium of the sealing jaw changes. If your lidding foil comes from a different supplier than the one used in the machine test, the bonding temperature range shifts.
Default settings are a starting point. Nothing more.
✔ How to Avoid It
During OQ, run a 3 × 3 design of experiment: three sealing temperatures (low, nominal, high) by three dwell times. Evaluate each combination with peel-force testing per ASTM F88 and CCI per ASTM F2338. Set the validated operating range at the intersection of passing results, with a safety margin of at least 5°C and 0.2 seconds from the acceptance boundary.

Sealing station with validated heat and pressure parameters — default supplier settings are not a substitute for OQ-qualified data.
Mistake 06
Incorrect Syringe Feeding Orientation — Plunger Rod Damage You Won’t Find Until In-Process Inspection
Prefilled syringe blister feeding orientation errors that cause plunger rod deformation or needle-shield displacement typically produce a 0.8–2.4% defect rate — low enough to miss on visual sampling, high enough to fail in-process inspection at the cartoning stage.
The stability report landed in March. The client had been running commercial production for six weeks. In-process visual inspection at the cartoning stage was flagging syringes with the needle shield rotated 15–22 degrees from the blister cavity centerline. The root cause was a feeding guide rail set 1.8 mm off-center from the cavity index position — a setup error from initial commissioning that nobody had verified against the acceptance criteria in the IQ protocol. Because nobody had written that acceptance criterion into the IQ protocol.
✔ How to Avoid It
Define feeding orientation tolerance in the URS as a measurable parameter: needle shield axis deviation ≤ ±3 degrees from cavity centerline, plunger rod tip clearance ≥ 2 mm from cavity wall. Include a go/no-go gauge check for feeding guide rail position in the IQ acceptance criteria. Run 500 cycles at commercial speed during OQ and inspect every 50th unit for orientation deviation.
Mistake 07
Incomplete GMP Validation Documentation — the Paper Trail That Regulators Actually Read
Incomplete or missing IQ/OQ/PQ documentation for syringe blister packaging systems is the single most common reason pharmaceutical facilities in emerging markets receive a Form 483 observation or WHO pre-qualification deferral during facility inspection — based on inspection records I’ve reviewed from clients in Nigeria, Indonesia, and Pakistan between 2021 and 2024.
The gap is predictable. A facility completes IQ and OQ. PQ gets compressed because the commercial launch date is fixed. The PQ report references three production runs, but two of them were at 70% commercial speed — not the intended 150 blisters/minute. The inspector asks for PQ data at commercial throughput. The answer is that it doesn’t exist. The facility is given six months to generate it. The launch slips.
Validation is not a formality. It’s the evidence that your packaging system performs as intended under the conditions it will actually operate. EU GMP Annex 15 requires PQ to be conducted at intended commercial conditions. WHO TRS 992 Annex 3 says the same. This is not ambiguous.
✔ How to Avoid It
Build the validation master plan before equipment is ordered. Define DQ, IQ, OQ, and PQ scope, acceptance criteria, and responsible parties in a single document. PQ must include a minimum of three consecutive runs at 100% intended commercial speed, with all critical quality attributes (seal integrity, orientation, print legibility, batch record completeness) documented to pre-defined limits. For GMP validation requirements in detail, review our dedicated compliance guide.

Forester Xiang
Founder, HIJ Machinery — 20 Years in Pharmaceutical Packaging
The seven mistakes above aren’t caused by bad machines. They’re caused by the structural gap between who specifies a syringe blister line and who runs it. Procurement teams optimize for unit cost. Engineering teams optimize for install timeline. QA teams arrive after the purchase order is signed. Nobody owns the system as a single integrated cGMP-compliant process until the FAT fails.
I won’t tell you the solution is simple. It involves your API profile, your target markets, your validation timeline, your facility infrastructure, and sometimes your relationship with the regulatory reviewer in your registration country. But the first step is always the same: treat the syringe blister line as a single validated system — forming, sealing, feeding, leaflet insertion, and cartoning — not a collection of individual machines that happen to sit next to each other.
At HIJ, we engineer the entire line around your specific syringe format from day one. What gets validated in FAT is exactly what runs in SAT. No surprises. No finger-pointing between vendors. That’s the only way I know to deliver a syringe blister packing machine that actually passes inspection.
Syringe Blister Packaging Mistake Impact — Cost & Timeline Comparison
The table below summarizes the seven syringe blister packaging mistakes, their typical detection point, and the cost impact when corrected at each stage. Detection at FAT is expensive. Detection post-commercial launch is potentially catastrophic.
| # | Syringe Blister Packaging Mistake | Typical Detection Point | Cost Impact if Caught at FAT | Cost Impact if Caught Post-Launch |
|---|---|---|---|---|
| 1 | Forming depth mismatch to barrel geometry | FAT (cycle 200–800) | $8,000–$22,000 mold rework | Batch rejection + $30,000+ tooling |
| 2 | Wrong lidding material, no Zone IVb data | Month 6–12 stability study | $15,000–$40,000 re-validation | $500K–$2M recall + registration delay |
| 3 | Multi-vendor PLC protocol conflict | FAT integration testing | 6–14 week FAT delay | Line downtime + emergency integration cost |
| 4 | CCI testing skipped / deferred | Dossier review / inspection | $1,200–$2,500 to add during OQ | 8+ month registration delay |
| 5 | Undertested sealing parameters | In-process inspection, Month 1–3 | $5,000–$12,000 re-OQ | Batch rejection + CAPA documentation |
| 6 | Wrong syringe feeding orientation | In-process, cartoning stage | $3,000–$8,000 fixture rework | 0.8–2.4% defect rate, AQL failure |
| 7 | Incomplete IQ/OQ/PQ documentation | Regulatory inspection | 4–6 weeks to complete PQ | Form 483 / WHO deferral, 6-month delay |
📋 Applicable Regulatory Standards
- EU GMP Annex 1 (2022) — Sterile medicinal products, container closure integrity
- EU GMP Annex 15 — Qualification and validation, PQ at commercial conditions
- WHO TRS 992 Annex 3 — GMP for pharmaceutical products, packaging validation
- ICH Q1A(R2) — Stability testing, Zone IVb conditions
- USP <1207> — Container closure integrity testing
- ASTM F2338 — Dye-ingress CCI test method
- ASTM F88 — Seal strength testing for flexible barrier materials
- ISO 11040-6 — Prefilled syringes, glass barrels, dimensional tolerances
Understanding the true cost of syringe blister packaging mistakes — early detection saves between 10x and 50x the correction cost versus post-commercial discovery.
⚠ Field Diagnostic — Validation & Integration Failure Patterns
Symptom: FAT passes, SAT fails on integration handshake. The forming station and cartoning unit communicate correctly in the controlled FAT environment (stable power, single-vendor oversight). At the client site, a voltage fluctuation of ±5V causes a PLC timeout that triggers an unscheduled stop. Neither vendor considers this their problem. Build a witnessed integration test at simulated site conditions — including power quality, ambient temperature, and operator interaction — into the FAT protocol. Don’t accept FAT completion until this is done.
Symptom: PQ batch records missing critical quality attribute data for 2 of 3 runs. Someone ran the first PQ batch as a “dry run” and didn’t complete the data collection form. Now you have one fully documented batch and two partial ones. Regulators count complete runs only. Start over. This is not negotiable under EU GMP Annex 15 or WHO TRS 992 Annex 3. The cost is four to eight weeks of production time. The prevention is a pre-PQ readiness checklist signed by QA before the first batch starts.
Symptom: Orientation defect rate increases after month 2 of commercial production. Guide rail wear. The feeding guide rail on a high-speed syringe blister line experiences friction-driven wear at the contact point with the syringe flange. At 150 cycles/minute, that’s 216,000 contact events per day. After 60 operating days, dimensional tolerance at the guide tip can drift by 0.5–1.2 mm — enough to reintroduce the orientation error you corrected at OQ. Add guide rail dimensional inspection to the monthly preventive maintenance schedule.
Pre-Procurement Checklist — 7 Questions to Answer Before Buying a Syringe Blister Packing Machine
Every one of the seven mistakes above is preventable with a structured pre-engineering review. Use this checklist before committing to equipment specification:
- Have you measured actual syringe barrel OD from a commercial batch sample (≥60 units)? Nominal data sheet dimensions are insufficient for mold cavity design.
- Do you have Zone IVb stability data for your target distribution geography? If any market is Zone IVb, your lidding material must have a verified MVTR specification below 0.5 g/m²/day.
- Is your entire line sourced from a single integration-responsible supplier? Multi-vendor PLC conflicts cannot be resolved after FAT has started. Assign integration accountability in the purchase contract before signing.
- Is CCI testing per ASTM F2338 included in your OQ protocol with pre-defined acceptance criteria? Deferring this to post-approval will cost you a regulatory submission delay of 6–12 months.
- Have you designed a sealing parameter DOE (3×3 minimum) that uses your actual commercial lidding foil lot? Supplier default settings are not validated parameters.
- Is syringe feeding orientation tolerance defined as a measurable IQ acceptance criterion? If it’s not in the IQ protocol, it won’t be checked — and it will drift.
- Does your validation master plan include PQ at 100% intended commercial speed for three consecutive runs? Partial-speed PQ data is not accepted under EU GMP Annex 15 or WHO TRS 992.
Frequently Asked Questions — Syringe Blister Packaging Mistakes & Quality Failures
❓ What is the most common cause of syringe blister packaging failures?
Forming depth mismatch between the thermoforming cavity and the actual syringe barrel outer diameter is the single most frequently occurring root cause of syringe blister packaging failures at FAT — appearing in approximately 35% of commissioning projects where the mold was designed from nominal data sheet dimensions rather than measured sample data. It produces cold-seal defects in 4–9% of blisters and requires mold rework costing $8,000–$22,000. The fix costs nothing at the design stage.
❓ Can I use standard PVC lidding for syringe blister packs going to tropical markets?
No — not for sterile prefilled syringes containing hygroscopic drug products distributed to WHO climatic Zone IVb markets. Standard PVC lidding passes 4–6 g/m²/day MVTR. Zone IVb markets (India, Southeast Asia, sub-Saharan Africa) require MVTR below 0.5 g/m²/day for most injectable biologics and lyophilized products. Using PVC without Zone IVb stability data is a registration risk and a potential recall trigger. Specify lidding material after confirming your distribution geography and running ICH Q1A(R2) Zone IVb stability.
❓ How long does syringe blister packaging validation (IQ/OQ/PQ) typically take?
A complete IQ/OQ/PQ validation for a syringe blister packing line typically takes 10–18 weeks from FAT completion to PQ sign-off, under normal conditions. IQ takes 3–5 days. OQ — including sealing parameter DOE and CCI testing — takes 3–6 weeks depending on laboratory turnaround. PQ requires three consecutive commercial-speed production runs, each fully documented, which adds 2–4 weeks. Facilities that defer CCI testing or compress PQ to below-commercial speed typically add 8–16 weeks of re-validation work. For the full requirements, review our guide on syringe blister packaging GMP compliance and validation.
❓ What is the real cost impact of syringe blister packaging mistakes?
The real cost depends on when the mistake is detected. Caught at design stage: near zero. Caught at FAT: $3,000–$40,000 in rework plus 6–14 weeks of timeline extension. Caught post-commercial launch: $500,000 to over $2 million in batch recalls, registration delays, and CAPA programs — plus the reputational cost in markets where your distributor relationships took years to build. The cost breakdown for each mistake type is detailed in our analysis of syringe blister packing machine total cost of ownership.
❓ Do multi-vendor syringe blister packaging lines create validation problems?
Yes. Multi-vendor lines consistently create integration gaps that appear at FAT — incompatible PLC protocols, mismatched signal timing, undefined reject handshake behavior. In HIJ’s commissioning records, multi-vendor lines averaged 6–14 weeks longer FAT timelines than single-supplier turnkey lines. The validation documentation burden also increases significantly because each machine requires a separate IQ, and the integration between machines must be validated as a separate scope item that no single vendor formally owns.
❓ Is container closure integrity testing required for all syringe blister packs?
Required for sterile drug products. USP <1207> and EU GMP Annex 1 (2022 revision) both mandate CCI testing for sterile packaging systems. For non-sterile syringe accessories or non-injectable products, the requirement depends on the drug product dossier and the regulatory authority in the target market. When in doubt, include CCI testing in OQ — it costs $1,200–$2,500 per validation run and eliminates a frequently cited dossier deficiency.
🎯 HIJ Machinery — Turnkey Syringe Blister Packaging
Avoid Every One of These Mistakes — Before the Line Is Built
HIJ engineers the entire syringe blister line — forming, sealing, feeding, leaflet insertion, cartoning — as a single validated cGMP system. What passes FAT is exactly what runs at SAT. No surprises. No vendor finger-pointing.