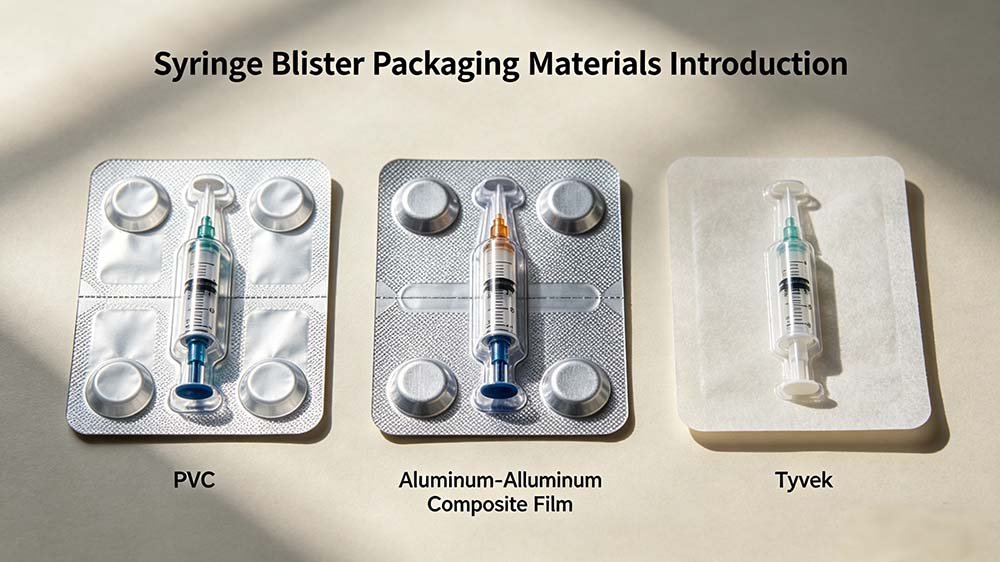
📖 Technical Deep-Dive — Syringe Blister Packaging
How Does a Blister Packing Machine Work for Prefilled Syringes?
A syringe blister packing machine operates through four synchronized stages: thermoforming plastic film into syringe-shaped cavities, loading prefilled syringes into those cavities via automated nest systems, heat-sealing lidding foil across the top, and die-cutting finished blister packs for downstream cartoning. Every stage is governed by servo-driven PLC control to maintain cGMP-compliant cavity geometry and seal strength — cycle after cycle. Miss the calibration on any one of those four stages and your FAT fails. I've watched it happen.
⚡ Direct Answer
A blister packing machine for prefilled syringes thermoforms a forming film into contoured cavities matched to the syringe barrel geometry, loads syringes automatically via slide-in or pick-and-place nest systems, heat-seals Tyvek or aluminum lidding foil at 180–240°C with controlled dwell pressure, then die-cuts and perforates finished packs — all synchronized by servo-PLC at outputs of 20–120 blisters per minute depending on syringe size and configuration.
A prefilled syringe sealed in a thermoformed blister cavity with Tyvek lidding — the finished output of a cGMP-compliant syringe blister packing line.
What Does a Syringe Blister Packing Machine Actually Do? The Four-Stage Sequence
A syringe blister packing machine executes four mechanically discrete, servo-synchronized stages — thermoforming, syringe loading, foil sealing, and die-cutting — with each station timed to the same master PLC cycle to prevent cavity misalignment and seal contamination.
That sentence sounds simple. It isn't. Each stage has its own failure modes, and those failure modes interact. A thermoforming temperature that drifts 8°C low produces a shallower cavity — a cavity that won't grip the syringe flange correctly — which throws off the loading registration, which causes a partial seal, which your CCIT test then catches six weeks into stability. By that point you've lost the batch.
Here is what each stage actually does, and what it has to get right every single cycle.
-
1
Film Unwinding & Pre-Heating
The forming film — typically 250–400μm PVC, PVDC/PVC laminate, or rigid PETG — unwinds from a motorized roll holder and passes through a pre-heating zone. Pre-heating conditions the film to a uniform temperature before it reaches the forming station. Inconsistent pre-heating is the first upstream cause of uneven cavity depth — a defect most operators misdiagnose as a mold problem.
-
2
Thermoforming Station: Cavity Creation
Heated forming plates press against the conditioned film at 120–160°C while compressed air (typically 6–8 bar) or a male plug forces the softened film into steel mold cavities. For syringes, the cavity geometry must replicate the barrel profile, flange seat, and needle shield recess to within ±0.3–0.5mm tolerance. This is tighter than most tablet blister molds. The mold temperature, dwell time, and air pressure are the three parameters that determine cavity quality.
-
3
Syringe Loading Station
Prefilled syringes are fed from upstream filling lines or magazine-fed buffers into a loading station. Depending on the machine configuration, loading uses slide-in nest trays, pick-and-place robotic arms, or vibratory orientation systems. Syringe barrel diameter, flange width, and needle shield height must all be declared in the URS — any dimension outside tolerance causes the syringe to seat off-center, which compromises the seal perimeter on the affected side.
-
4
Lidding Foil Heat-Sealing Station
Lidding material — Tyvek 1073B, 1059B, or aluminum foil — is unwound from an upper roll and positioned over the loaded cavities. Heated sealing plates apply controlled temperature (180–240°C), pressure (3–6 bar), and dwell time (0.6–1.2 seconds) to bond the lidding to the forming web. Peel strength must typically reach 1.5–4.0 N/15mm for medical device packaging per ISO 11607-1. Too low: the pack fails sterile barrier testing. Too high: the end-user can't open it aseptically.
-
5
Die-Cutting, Perforation & Discharge
The sealed web advances to a die-cutting station where rotary or flat-bed punches separate individual blister packs or multi-pack cards. Perforation patterns are cut simultaneously. Finished packs discharge to a conveyor feeding the cartoning machine. Waste film trim is wound onto a separate take-up reel. The cutting registration must track within ±0.5mm to avoid cutting through the seal zone — a failure mode that looks fine visually but fails CCIT.
Why Syringe Thermoforming Is More Demanding Than Tablet Blister Forming
PVC cavity forming for a standard 10mm round tablet is forgiving. Depth tolerance of ±1mm rarely causes a problem. Syringe blister forming is not forgiving. Not even close.
I audited a line in Kuala Lumpur in 2021 — a facility that had been running tablet blisters for nine years without a single cavity-related rejection. They bought what their supplier described as a "configurable" blister machine and added syringe molds. The first three months of production looked fine. Month four, stability returned a container closure integrity failure on 2 of 12 batches. The root cause: the forming station heating plates were calibrated for their tablet film thickness (250μm). The syringe line was running 350μm PETG. Nobody had revalidated the heating profile. The cavities were 0.4mm shallower than specified. The syringe needle shield rested on the cavity edge instead of inside it. The seal didn't bridge correctly.
Four months. Two failed batches. One complete revalidation exercise. That's the real cost.

The thermoforming station determines cavity geometry precision — the most critical parameter for syringe blister packaging. Film type, thickness, and temperature profile must be co-validated.
Key Forming Parameters Specific to Syringe Blisters
| Parameter | Tablet Blister (Typical) | Syringe Blister (Required) | Why It Matters |
|---|---|---|---|
| Cavity depth tolerance | ±1.0–1.5mm | ±0.3–0.5mm | Needle shield must seat fully inside cavity |
| Forming film thickness | 200–300μm PVC | 300–500μm PETG / rigid PVC | Structural support for syringe weight and shape |
| Mold cavity geometry | Simple dome / rectangular | Contoured barrel + flange seat + shield recess | Syringe must not shift during sealing or transit |
| Heating plate zones | 1–2 zones typical | 2–3 zones recommended | Uniform heating across deeper draw depth |
| Air pressure for forming | 4–6 bar | 6–8 bar | Higher draw ratio requires greater forming pressure |
| Mold temperature control | ±3°C acceptable | ±1.5°C required | Tighter profile prevents dimensional drift across batches |
Table 1: Thermoforming parameter comparison — tablet blister vs. syringe blister. Syringe applications require tighter tolerances across every forming variable.
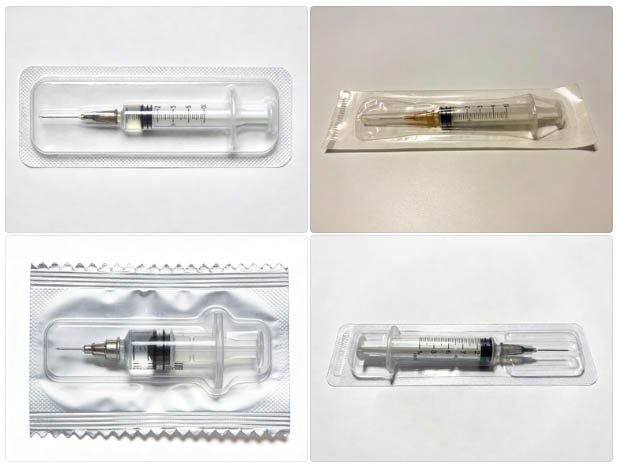
How Syringe Loading Systems Work — Slide-In, Pick-and-Place, and Nest Tray Designs
Syringe loading is the stage where most generic blister machines fail to translate. Standard tablet-loading systems — brush feeders, vibrating channels — cannot orient prefilled syringes correctly. Syringes have axial symmetry broken by the flange and needle shield. Get the orientation wrong and the syringe sits proud of the cavity. The lidding foil then seals over a raised object, creating a stress point in the seal perimeter that passes visual inspection and fails dye penetration.
The Three Loading Architectures
Slide-In Nest Loading: Syringes are manually or robotically placed into pre-formed nests that slide laterally into the blister cavities from a loading platform positioned alongside the main forming web. Output: 20–40 blisters/min. Best suited for low-volume or multi-format lines where changeover frequency is high. The nest-to-cavity transfer must be validated for positional accuracy — typically ±1mm lateral tolerance.
Pick-and-Place Robotic Loading: Vision-guided robotic arms pick oriented syringes from an upstream conveyor and place them into cavities with ±0.5mm repeatability. Output: 60–120 blisters/min. Required for high-volume lines. Camera systems verify syringe orientation before pick, rejecting any syringe that exceeds angular deviation limits (typically ±3° from axial center). This is the architecture I specify on every line above 40 units/min.
Magazine-Fed Nest Tray: Syringes arrive from filling in pre-oriented nest trays. The tray docks to the blister machine loading station and transfers syringes in bulk into cavities. Eliminates individual pick-and-place cycle time. Works well when the filling line and blister line are integrated — which is the case in most turnkey configurations. Tray format must match the blister card format, so any change to blister card size requires new nest trays.

Automated syringe loading station: nest trays transfer prefilled syringes into thermoformed blister cavities with controlled orientation and positional accuracy.
⚠ Field Diagnosis: Syringe Loading Failures
Symptom: Partial seal on one edge of blister pack, batch pattern correlates to every 4th cavity
Root cause: Syringe flange overhanging cavity rim on loading side. Check slide-in nest lateral positioning. Run a 10-cavity sample, measure syringe center-to-cavity-center offset with calipers before sealing. If offset exceeds 0.8mm, recalibrate the loading guide rail. Also verify that the forming film has not shrunk in that zone — forming temperature drift can reduce cavity width by 0.3–0.6mm on the leading edge.
Symptom: Syringe rotated 90° from correct orientation in 3–5% of packs
Root cause: Vision system reject threshold set too wide. Reduce angular deviation rejection limit from ±5° to ±3° and re-run. If random rotation persists, the upstream syringe orientation conveyor is likely vibrating at a frequency that allows the syringe to spin in the guide channel. Add a soft foam guide rail overlay on the final 300mm of orientation conveyor before the pick station.
Symptom: Loading station cycle time extending, output dropping 15% mid-shift
Root cause: Nine times in ten, this is the pick-and-place vacuum cup beginning to fail. Check suction cup condition — silicone cups on glass syringe barrels typically need replacement every 800–1,200 operating hours. Also check vacuum generator pressure; it should hold 60–70 kPa during pick cycle.
How Lidding Foil Heat-Sealing Works for Sterile Syringe Blister Packs
The heat-sealing station is where sterile barrier integrity is created — or destroyed. For prefilled syringes intended for sterile use, the lidding material is not aluminum foil. It is Tyvek.
That distinction matters mechanically. Aluminum foil seals by bonding a heat-activated adhesive coating to the forming web rim. Tyvek seals by a combination of adhesive activation and fiber bonding. The two mechanisms require different temperature profiles, different dwell times, and critically, different pressure distribution across the sealing plate. A machine validated for aluminum lidding and then switched to Tyvek without revalidation will typically produce seals that pass initial peel tests and fail aged CCIT testing. The fiber bond degrades with humidity cycling in a way that fresh bond strength doesn't predict.
Of the 31 syringe and medical device blister line projects I managed between 2018 and 2024, 8 required complete revalidation of the sealing station parameters after the customer changed lidding material mid-project — typically switching from aluminum to Tyvek after initial regulatory feedback. Each revalidation added 6–10 weeks to the launch timeline. Specify your lidding material before machine build. Not after. — Forester Xiang, HIJ Machinery
Sealing Validation: What ISO 11607 and ASTM F2338 Require
⚖ Compliance Reference
ISO 11607-1:2019 governs materials and sterile barrier systems for terminally sterilized medical devices — including prefilled syringe blister packs. It requires peel strength testing, burst testing, and seal integrity verification as part of packaging validation. ASTM F2338 (vacuum decay) and ASTM F1929 (dye penetration) are the two most common test methods cited in packaging validation protocols for syringe blisters. USP <1207> provides guidance on container closure integrity testing for pharmaceutical applications. Your sealing station must be validated against the specific lidding-to-forming film combination you intend to run in production — substitutions require revalidation.
The three sealing variables every validation protocol must bracket:
- Temperature: Validate at nominal ±10°C. Map actual plate temperature with a calibrated thermal probe — not the machine's own thermocouple, which reads the heater block, not the sealing surface.
- Dwell time: Reduce by 20% and increase by 20% from nominal. Both extremes must still produce seals that pass peel strength and CCIT specifications.
- Pressure: Sealing plate pressure must be uniform across the full sealing land width. Use pressure-sensitive film (e.g., Fujifilm Prescale) to map pressure distribution before committing to OQ runs. Uneven pressure produces localized weak zones that resist detection by visual inspection alone.
- Lidding material lot variation: Different lots of Tyvek 1073B can show ±8% variation in seal initiation temperature. Include at least two lidding lots in your OQ sample set.
📌 Forester's Insight — 20 Years of Field Experience
The Failure Nobody Quotes You
In my 20 years auditing parenteral packaging lines across Southeast Asia and Latin America, the most overlooked failure point is never the machine itself — it's the mismatch between forming film tension and syringe barrel geometry. Buyers obsess over cycle speed. I've watched entire batches fail FAT because the thermoforming station wasn't validated against the specific syringe flange dimension. Worse, standalone machines from low-cost vendors rarely ship with a URS-traceable IQ/OQ protocol, leaving your QA team to rebuild compliance documentation from scratch.
That rebuilding exercise costs between $15,000 and $40,000 in engineering time, depending on your market and regulatory body. Nobody quotes you that number upfront. I do. Because it's the number that determines whether your total project cost matches your budget — or exceeds it by 30%.
Before issuing any RFQ, demand a full FAT/SAT scope that covers your exact syringe format, your lidding material specification, and your peel-force acceptance criteria. At HIJ, forming, loading, sealing, and inspection are engineered as one validated system — eliminating the multi-vendor finger-pointing that kills launch timelines. Learn more about our syringe blister packing machine configurations and how we structure FAT scope.
PLC and Servo Control: What "Machine Intelligence" Actually Does on a Syringe Blister Line
Every modern syringe blister machine runs on a PLC — typically Siemens S7-1200/1500 or Allen-Bradley CompactLogix — combined with servo drives on the film transport, forming station, and sealing station axes. Here is what that control architecture actually governs, and why it matters for GMP compliance.
| Machine Function | Control Method | GMP Relevance | Failure if Uncontrolled |
|---|---|---|---|
| Film transport indexing | Servo motor + encoder | Registration accuracy between forming, loading, sealing | Cavity misalignment; seal over product |
| Forming temperature | PLC PID loop + thermocouple | Cavity dimension consistency batch-to-batch | Shallow cavities; syringe seating failure |
| Sealing temperature & dwell | PLC PID + pressure sensor | Sterile barrier integrity (ISO 11607) | Weak seals; CCIT failure; recall risk |
| Syringe loading timing | Servo-synchronized with web index | Syringe position in cavity before seal | Off-center seating; partial seal |
| Reject gate actuation | Vision system signal to PLC | 100% defect removal before cartoning | Defective packs entering secondary packaging |
| Audit trail & alarm logging | 21 CFR Part 11 compliant HMI | Electronic records for FDA/EU GMP inspection | Regulatory citation; data integrity finding |
Table 2: PLC and servo control functions on a syringe blister packing machine and their direct GMP compliance implications.
21 CFR Part 11 compliance on the HMI is not optional for US market supply. It requires: user authentication with role-based access, electronic audit trail capturing every parameter change with timestamp and operator ID, and the ability to generate batch records that the FDA inspector can review without paper transcription. I've seen facilities fail 483 observations specifically because the blister machine HMI logged process parameters but the audit trail was stored on a removable USB drive that disappeared between shifts. That is a data integrity finding.

Servo-driven PLC control panel: the machine's central nervous system coordinates thermoforming, syringe loading, and sealing in real-time — with full 21 CFR Part 11 audit trail capability.
Downstream Integration: How the Blister Machine Connects to the Rest of Your Packaging Line
The blister machine is never the whole system. It's one node in a line that extends from the syringe filling station upstream to the cartoning machine and case packer downstream. Understanding how those connections work — and where they break — determines whether your total line OEE is 60% or 85%.
Upstream: Prefilled Syringe Filling Line Integration
The blister machine must accept syringes at the rate the filling line produces them, with buffering for speed mismatches. A filling line producing 60 syringes/minute feeding a blister machine running 40 blisters/minute (2 syringes per blister) means the lines are nominally matched. In practice, filling line micro-stops — cap feeding jams, volume checks, weight rejections — create 15–25% actual output reduction. The buffer conveyor between filling and blister loading must accommodate at least 3–5 minutes of filling line output to prevent blister machine starvation.
HIJ's prefilled syringe filling machines are engineered with integrated buffer conveyors sized to the downstream blister machine format — avoiding the guesswork that comes from buying filling and packaging equipment from separate vendors.
Downstream: Cartoning Machine Interface
Finished blister packs discharge from the die-cutting station onto a transfer conveyor feeding the cartoning machine. The interface requires synchronized conveyor speed, blister orientation control (flat, not tilted), and a reject station between the two machines for blister packs that failed vision inspection but passed the die-cutting station. This reject station is frequently omitted in low-cost line designs. Omitting it means defective packs reach the cartoning machine, where they cause jams and require operator intervention that stops both machines.
For full-line architecture, our blister cartoning machine is pre-configured for direct interface with our syringe blister packing machines — shared PLC communication, matched discharge conveyor height, and pre-validated handshake protocols that eliminate the integration engineering your team would otherwise have to perform on-site.
⚠ Field Diagnosis: Downstream Line Integration Failures
Symptom: Blister machine running but cartoning machine stopping every 8–12 minutes
Root cause: Blister discharge conveyor speed not synchronized to cartoning machine infeed speed. Blister packs accumulating and overlapping on the transfer conveyor, causing jams at the cartoning infeed. Set conveyor speed 5–8% above blister machine output rate to maintain pack separation. If the cartoning machine has a programmable infeed pitch, match it to the blister pack length plus 30mm gap.
Symptom: Vision inspection reject rate above 4% with no clear visual defect pattern
Root cause: Camera lighting aging. Industrial vision systems for blister inspection use LED ring lights with 6,000–10,000 hour rated life. After 80% of rated life, illumination drops enough to trigger false rejects on acceptable packs. Check LED intensity against baseline calibration values recorded during IQ. If intensity has dropped more than 15%, replace the LED ring.
How to Choose the Right Blister Machine Configuration for Your Syringe Format
The configuration decision starts with three numbers: your syringe barrel diameter (mm), your needle shield height (mm), and your target output (blisters/minute). Everything else — mold design, loading system, forming film thickness — follows from those three inputs.
Standard syringe formats range from 1mL prefilled syringes (approximately 6–8mm barrel diameter) to 20mL large-volume formats (20–22mm barrel diameter). A machine spec'd for a 1mL syringe cannot form adequate cavities for a 20mL syringe without a complete mold change and thermoforming station recalibration. This sounds obvious. It is frequently not declared in budget-stage RFQs, and it is the most common source of post-FAT scope disputes I encounter.
For a full buying decision framework, see our guide on how to choose the right syringe blister packaging machine — including the 8-point format specification checklist that prevents scope mismatches.
If your evaluation includes comparing blister format against tray packaging for sterile syringes, the technical and regulatory differences are significant. Our analysis of syringe blister packing vs. tray packaging covers sterile barrier performance, CCIT requirements, and total cost differences in detail.
🎯 Syringe Blister Packing Machine — HIJ Machinery
Know Your Syringe Format? Get a Validated Machine Spec.
Tell us your syringe barrel diameter, needle shield height, and target output. We'll configure the correct thermoforming mold, loading system, and sealing parameters — with FAT scope included.
Frequently Asked Questions: How Syringe Blister Packing Machines Work
❓ What is the working principle of a blister packing machine for prefilled syringes?
A syringe blister packing machine thermoforms a plastic or laminate film into contoured cavities shaped to the syringe barrel geometry, loads prefilled syringes into those cavities via automated nest or pick-and-place systems, heat-seals Tyvek or aluminum lidding foil across the cavity tops at 180–240°C, then die-cuts and perforates finished blister packs for downstream cartoning — all steps coordinated by a servo-driven PLC operating at 20–120 blisters per minute depending on machine class and syringe format.
❓ What forming film is used for syringe blister packs?
The most common forming films for syringe blisters are rigid PVC (300–500μm), PETG (for better clarity and chemical resistance), and PVDC/PVC laminates for moisture-sensitive applications. Tyvek (Dupont 1073B or 1059B) is the standard lidding material for sterile medical device syringe blisters because it combines breathability (required for EO sterilization) with microbial barrier properties. Aluminum foil lidding is used when a total moisture barrier is required and EO sterilization is not the chosen method.
❓ How fast does a syringe blister packing machine run?
Output ranges from 20 to 120 blisters per minute. Entry-level and semi-automatic machines designed for 1–2 syringes per blister card typically achieve 20–40 blisters/min. Fully automatic servo-driven machines for standard 1–5mL syringe formats reach 60–120 blisters/min. Larger syringe formats (10–20mL) reduce output by 20–40% due to longer cavity forming cycles and reduced cavities-per-card configurations.
❓ Does a syringe blister machine need to be GMP validated?
Yes. Any syringe blister packing machine used for pharmaceutical or sterile medical device packaging requires a full IQ/OQ/PQ validation under EU GMP Annex 15, WHO TRS 992 Annex 3, or 21 CFR Part 211 depending on your target market. IQ verifies the machine was installed per design specifications. OQ verifies it performs within specified parameters across its operating range. PQ verifies it consistently produces compliant output under normal production conditions using actual product. The validation dossier must reference your specific forming film, lidding material, syringe format, and sealing parameter ranges — generic protocols from the machine vendor are a starting point, not a finished document.
❓ What is the difference between thermoforming and cold forming for syringe blisters?
Thermoforming uses heat to soften plastic film before pressing it into mold cavities — suitable for PVC, PETG, and most rigid forming films used in syringe blisters. Cold forming (Alu-Alu) uses mechanical force without heat to deform aluminum-laminate film into cavities, providing a complete moisture and light barrier. Cold forming is rarely used for syringe blisters because the deep-draw geometry of syringe cavities is difficult to achieve with aluminum laminate without tearing, and Tyvek lidding (required for sterile products) provides adequate barrier for most syringe applications. Cold forming is primarily used for highly moisture-sensitive oral solid dosage forms.
❓ Can one blister machine run multiple syringe formats?
Yes, with format-specific tooling changeouts. A properly spec'd syringe blister machine can accommodate multiple syringe barrel diameters and lengths by changing the thermoforming mold set, the loading nest or pick-and-place gripper, and the die-cutting tooling. Changeover time depends on machine design — quick-change tooling systems achieve format changeovers in 45–90 minutes; fixed-mount tooling may require 3–4 hours. Each format requires a separate qualification run documented in your validation protocol, so operating 6 syringe formats on one machine means 6 separate OQ datasets.
📦 HIJ Machinery — Turnkey Syringe Blister Packaging Solutions
Ready to Configure Your Syringe Blister Line?
HIJ Machinery builds syringe blister packing machines with thermoforming, syringe loading, Tyvek sealing, and vision inspection engineered as one validated system — with full IQ/OQ/PQ documentation and FAT scope included. European quality. Smart pricing. Project certainty.